Short-day treatment in late summer reduces the chilling requirement in Norway spruce seedlings
Partanen J., Häkkinen R., Viherä-Aarnio A., Stenvall N., Hänninen H. (2026). Short-day treatment in late summer reduces the chilling requirement in Norway spruce seedlings. Silva Fennica vol. 60 no. 2 article id 25053. https://doi.org/10.14214/sf.25053
Highlights
- Short-day treatment reduces the chilling requirement and the time to bud burst in Norway spruce seedlings
- Forcing photoperiod does not affect the time to bud burst of short-day treated seedlings
- In natural outdoor conditions, the short-day treatment advances the bud burst slightly.
Abstract
Artificial shortening of photoperiod with short-day (SD) treatment in late summer is a common measure in forest nurseries to advance the growth cessation and increase the frost hardiness of seedlings in the autumn. We conducted an experimental study of the effect of SD treatment (12 h) in the preceding summer and the photoperiod (16 h, 8 h) prevailing in the regrowth test under forcing conditions. The effect of these factors on the chilling requirement in second-year Norway spruce Picea abies (L.) Karst. seedlings was studied. Half of the seedlings were first subjected to a three-week SD treatment between 11 July and 1 August 2005, whereas the other half were used as the control group. The seedlings were first exposed to chilling in natural conditions. Subsequently, samples of the seedlings were transferred at intervals between September and May to a regrowth test in growth-promoting forcing conditions in a greenhouse. Additionally, we observed the bud burst of the seedlings in natural conditions during the next spring. Our main result was that SD treatment reduced the chilling requirement in the seedlings. Long photoperiod (16 h) in the forcing conditions also reduced the chilling requirement. The buds of the SD-treated seedlings burst earlier than those of the control seedlings and generally at almost the same time in both photoperiods. This suggests that the photoperiod prevailing in the forcing conditions has no additional delaying or advancing effect on the bud burst of SD-treated seedlings. In natural outdoor conditions, the SD treatment in the preceding summer advanced the springtime bud burst slightly.
Keywords
bud burst percentage;
days to bud burst;
rest;
endodormancy release;
forcing, quiescence
-
Partanen,
Natural Resources Institute Finland (Luke), Production Systems, Juntintie 154, FI-77600 Suonenjoki, Finland
 https://orcid.org/0009-0007-1782-4283
E-mail
ext.jouni.partanen@luke.fi
https://orcid.org/0009-0007-1782-4283
E-mail
ext.jouni.partanen@luke.fi
-
Häkkinen,
Natural Resources Institute Finland (Luke), Production Systems, P.O. Box 2, FI-00791 Helsinki, Finland
 https://orcid.org/0009-0007-1687-1045
E-mail
risto.hakkinen@yahoo.com
https://orcid.org/0009-0007-1687-1045
E-mail
risto.hakkinen@yahoo.com
-
Viherä-Aarnio,
Natural Resources Institute Finland (Luke), Production Systems, P.O. Box 2, FI-00791 Helsinki, Finland
 https://orcid.org/0000-0002-1777-637X
E-mail
annelivihe@outlook.com
https://orcid.org/0000-0002-1777-637X
E-mail
annelivihe@outlook.com
-
Stenvall,
Natural Resources Institute Finland (Luke), Production Systems, P.O. Box 2, FI-00791 Helsinki, Finland
 https://orcid.org/0000-0002-9447-8712
E-mail
niina.stenvall@luke.fi
https://orcid.org/0000-0002-9447-8712
E-mail
niina.stenvall@luke.fi
-
Hänninen,
State Key Laboratory for Development and Utilization of Forest Food Resources, Zhejiang A&F University, 666 Wusu Street, Hangzhou 311300, PR China
 https://orcid.org/0000-0003-3555-2297
E-mail
hhannin@zafu.edu.cn
https://orcid.org/0000-0003-3555-2297
E-mail
hhannin@zafu.edu.cn

Received 20 October 2025 Accepted 24 March 2026 Published 16 April 2026
Views 6279
Available at https://doi.org/10.14214/sf.25053 | Download PDF

Supplementary Files
1 Introduction
Air temperature is the main environmental factor that regulates the timing of the vegetative bud burst of seedlings and trees growing in boreal and temperate regions with seasonally alternating climates. The timing of bud burst in Norway spruce Picea abies (L.) Karst. is mainly a response to the accumulation of chilling and temperature sum (Viherä-Aarnio et al. 2014 and references therein). Short photoperiods cause growth cessation, bud set, and dormancy induction in late summer and early autumn (Wareing 1956; Vaartaja 1959; Ekberg et al. 1979; Junttila 2007), but the timing of these developmental events is also affected by several interactions with air temperature (Koski and Selkäinaho 1982; Koski and Sievänen 1985; Partanen and Beuker 1999; Tanino et al. 2010).
After growth cessation and bud set, the buds enter a state of endodormancy (Lang et al. 1987; synonymous to rest; Fuchigami et al. 1982). Endodormancy release (synonymous to rest break; Fuchigami et al. 1982) means that the growth-arresting physiological conditions inside the buds are removed by chilling temperatures (e.g., −5 < T < +10 °C). After rest completion, i.e., meeting the chilling requirement of endodormancy release, buds attain full ontogenetic competence, but bud burst and growth onset are prevented by unfavourable environmental conditions, typically low, non-growth-promoting air temperatures prevailing in winter. This dormancy category is called ecodormancy (Lang et al. 1987; synonymous to quiescence; Fuchigami et al. 1982). Bud burst takes place as a result of prolonged exposure to sufficiently long periods of sufficiently high temperatures (e.g., T > +5 °C) (Romberger 1963; Sarvas 1974).
Mean air temperatures are projected to rise with the predicted climate change in the future. Daily mean temperatures will drop below 10 ℃ later in the autumn and temperatures above 0℃ will occur earlier in the spring (Ruosteenoja et al. 2020). This may have important impacts on forest ecosystems. High autumn temperatures may interfere with the development of frost hardiness in many tree species (Hänninen 2016; Wallin et al. 2017). Moreover, warm spells in winter and spring may trigger premature deacclimation and result in frost injuries (Augspurger 2013; Ma et al. 2019; Svystun et al. 2021).
In experimental conditions, photoperiod affects endodormancy release though in natural conditions it has been considered to have only a minor effect in comparison with that of the air temperature. Long days have been observed to partially compensate for the lack of chilling in several tree species, including Norway spruce (Nienstaedt 1967; Worrall and Mergen 1967). Moreover, experimental studies have shown that the timing of bud burst in Norway spruce is affected by both the direction of change in photoperiod (Partanen et al. 1998) and the accumulated duration of light period (Partanen et al. 2001), suggesting that day length may have a greater influence on the timing of bud burst than the mere compensation for the lack of chilling. If the forcing takes place in long days (16–20 h) then Norway spruce seedlings do not have an absolute chilling requirement for responding to normal forcing temperatures (Nienstaedt 1967; Worrall and Mergen 1967; Søgaard et al. 2008; Viherä-Aarnio et al. 2014). If the chilling requirement has not been met, however, Norway spruce cannot show bud burst in short photoperiods (Nienstaedt 1967; Worrall and Mergen 1967).
In northern latitudes, tree nurseries producing seedlings for forestry often resort to shortening of photoperiod artificially in late summer so as to produce well-hardened seedlings (Dormling et al. 1968; Heide 1974a; Colombo et al. 2001 and references therein; Wallin et al. 2017; Fløistad and Granhus 2019). SD-treated seedlings do not require frost protection in nurseries during autumn and are well-hardened for freezer storage in late autumn (Riikonen and Luoranen 2020; Riikonen et al. 2023). Moreover, the risk of frost damage in SD-treated seedlings decreases after autumn planting (Dormling et al. 1968; Heide 1974a; Colombo et al. 2001 and references therein). The artificial shortening of photoperiod with SD treatment in forest nurseries during a 2–3-week period in late summer stops height growth and advances bud formation, which also hastens the frost hardening in early autumn (Dormling et al. 1968; Colombo et al. 2001 and references therein; Konttinen et al. 2003; Fløistad and Granhus 2010). Riikonen and Luoranen (2020) found that an SD treatment started in mid-July increased bud frost hardiness in October but no longer in November. According to Riikonen et al. (2023), warm spells followed by low temperatures in boreal areas may predispose SD-treated Norway spruce seedlings to frost damage in the winter (i.e., in March) but not in the late autumn (i.e., October and November). Moreover, the SD treatment advances the bud burst in the next spring (Heide 1974b; Sandvik 1980; Fløistad and Granhus 2010). Luoranen and Sutinen (2017) found that the buds of SD-treated seedlings have fewer and shorter protective bud scales, which may be a partial reason for the early bud burst of SD-treated seedlings. On the other hand, it has been observed that in many tree species, including Norway spruce, relatively high temperatures during SD exposure and bud formation lead to a deeper dormancy, thus increasing chilling requirement for dormancy release and the high-temperature requirement for bud burst (Dormling 1989; Heide 2003; Søgaard et al. 2008; Kalcsits et al. 2009; Malyshev 2020; Beil et al. 2021; Garrigues et al. 2023). In this way, contrary to the notion stated above, relatively high temperatures during the SD treatment could delay, not advance, bud burst in the next spring. Hänninen (2016) refers to this phenomenon by the term quantitative dormancy induction.
As discussed above, the effects of an SD treatment in the growing season on bud burst in the next spring have been documented well in earlier studies. However, the possible effects of the photoperiod applied in the experimental forcing treatments of chilling-forcing experiments have not been studied previously in connection with SD treatment in the growing season. The effect of photoperiod needs to be studied, for photoperiod varies globally between different locations but remains the same from year to year in each location. Thus, it is not dependent on the climate change and variation in weather conditions. To fill this knowledge gap, we conducted an experimental study of the effects of an SD treatment in the preceding summer and of the photoperiod in experimental forcing conditions in a greenhouse (8 h vs. 16 h) on the chilling requirement of second-year Norway spruce seedlings. We transferred the seedlings from natural outdoor chilling conditions to forcing conditions in several batches between September and May. We observed the bud burst of the SD-treated and the control seedlings not only in the forcing conditions but also in the natural conditions of the next spring. Our hypothesis was that both the SD treatment in late summer and the long photoperiod in forcing conditions reduce the chilling requirement of the seedlings and advance the bud burst.
2 Material and methods
2.1 Seedling material
The second-year seedlings were grown in the nursery of the Suonenjoki Research Unit (62°39´N, 27°03´E; 142 m a.s.l.) of the Natural Resources Institute Finland (Luke), by using seeds collected from seed orchard number 177 Sairila in Hartola (61°34´N, 26°05´E; 100 m a.s.l.). The seed orchard includes 76 grafted clones of Norway spruce, all of them having their origin in a restricted geographical area in the interior of southern Finland (Nikkanen et al. 1999). The seedlings were produced with a mixture of this seed material. The seeds were sown on 28 May 2004 in Plantek 81F (BCC, Landskrona, Sweden) trays (81 cells per tray, 549 cells m–2, cell volume 85 cm3) filled with fertilised Kekkilä (Eurajoki, Finland) M6 W peat for forest nurseries (the symbol W indicates inclusion of non-ionic wetting agent). The seedlings were grown for two growing seasons by using the standard nursery practices applied in Finland (Juntunen and Rikala 2001; Lilja et al. 2010). In 2004, the seedlings were grown on a growth sheet in an unheated plastic greenhouse, and in 2005, in an outdoor growing area. In both years, the seedlings were fertilised with a commercial fertiliser (Kekkilä Pot Plant Superex, N 19%, P 4% K 20%) dissolved in water. During the first growing season, the seedlings were fertilised once with 8 g m–2 of the commercial fertiliser. During the second growing season, when the SD treatments were carried out, the SD-treated and the control seedling were fertilised with a total of 60 g m–2 between 2 June and 8 July, applied in several dozes varying between 5 and 10 g m–2, and on 2 August, with 10 g m–2. During the three-week SD (12 h) treatment from 11 July to 1 August, the SD-treated seedlings were fertilised with a total of 27 g m–2 and the control seedling with a total of 49 g m–2, applied in several dozes varying between 5 and 10 g m–2. The control seedlings got slightly more fertilisation (22 g m–2) that time, which may have caused a slight increase in their nutrient reserves. However, it was not possible to evaluate the possible effect of this in the present study.
On 30 August 2005, the SD-treated and the control seedlings were transported to the Punkaharju Research Unit (61°48´N, 29°19´E) of the Natural Resources Institute Finland, Luke. Between 5 September and 7 September, the seedlings were transplanted from the Plantek 81F trays to Soparco (Le Musset, Condé-sur-Huisne, France) 7 cm × 7 cm × 8 cm (300 cm3; outside measures) plastic pots filled with fertilised Kekkilä M6 peat for forest nurseries. After transplanting the seedlings, the pots were put in polystyrene boxes to shelter the roots during the winter and moved to natural outdoor conditions. The storage area was enclosed with a 60-cm-high metal net to protect the plant material against voles.
2.2 Experimental design
During their second growing season, half of the seedlings were subjected to a three-week SD (12 h) treatment at the Suonenjoki Research Unit from 11 July to 1 August 2005, whereas the other half were used as a control group (Fig. 1). In the SD-treatment, the blackout (i.e., the dark period between 8 pm and 8 am) was implemented by means of a blackout curtain in the outdoor growing compound. The control seedlings were grown under natural photoperiod, which decreased from 19 h 11 min on 11 July to 18 h 12 min on 1 August. After the SD-treatment, all seedlings were kept under natural conditions at Suonenjoki until the start of the chilling-forcing experiment.
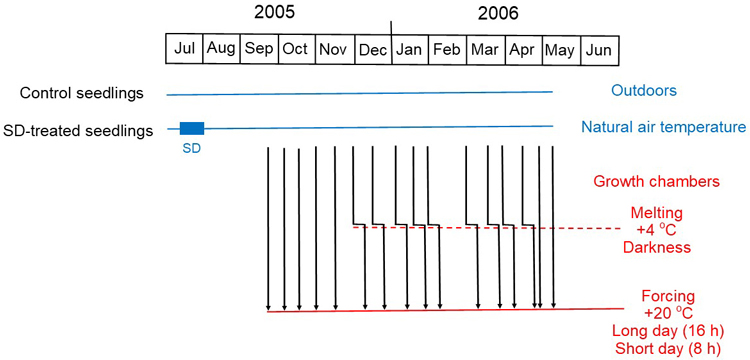
Fig. 1. Experimental design of the study. In the summer of 2005, the experimental Norway spruce seedlings were divided into two groups: half of the seedlings were kept all the time in natural conditions outdoors (Control seedlings), whereas the other half was exposed outdoors with black curtains to a short-day (12 h) treatment from 11 July to 1 August (SD-treated seedlings). The short-day treatment is indicated by the blue box on the left-hand side of the figure. During the next overwintering period seedlings from both groups were transferred on 16 occasions from outdoors to forcing conditions in growth chambers (vertical arrows, for the exact dates, see Table 1), thus exposing the seedlings in the different transfers to different durations of natural chilling. Forcing took place both under long-day (16 h) and short-day (8 h) conditions, air temperature was +20 °C in both. In the forcing conditions the occurrence (bud burst percentage, BB%) and timing (days to bud burst, DBB) of bud burst was examined. The explaining factors were the short-day treatment in the preceding summer, transfer time from the chilling conditions to the forcing conditions, and the day length in the forcing conditions. In nine transfers in the winter the seedlings were frozen, so before transferring them to the forcing conditions they were carefully melted for one week in darkness under +4 °C (angular arrows in the middle of the figure). In the analysis of the results the one week under +4 °C was considered as chilling, so that the time of the second transfer, i.e., the transfer from +4 °C to the forcing conditions, was used as the transfer time.
In 2005 and 2006, a chilling-forcing experiment was carried out at the Punkaharju Research Unit between 20 September and 31 May. The SD-treated and the control seedlings were first exposed to chilling in natural conditions (Fig. 1). Then seedlings of both SD- and control treatment were transferred to a regrowth test in two different forcing conditions in two respective greenhouse compartments (Fig. 1). There were 16 transfers at intervals of two to four weeks. Temperature in both greenhouse compartments was constant +20 °C. Day length in one compartment was 8 h and in the other 16 h (Fig. 1). In each of the 16 transfers, both the SD-treated and the control seedlings were randomised in regard to the two respective photoperiods in the two greenhouse compartments. The first samples were moved to forcing conditions on 20 September 2005 and the last ones on 9 May 2006 (Table 1). At each of the 16 transfers, ten SD-treated and ten control seedlings were sampled from the chilling conditions to each of the two forcing conditions with the two respective photoperiods (n = 10) (Fig. 1). Meanwhile, twenty SD-treated seedlings and twenty control seedlings were kept outdoors for observing bud burst in natural conditions in the next spring.
| Table 1. Dates when the seedlings were transferred in 2005–2006 to a regrowth test under forcing conditions in a greenhouse, either directly from natural outdoor conditions or after cautious melting in preforcing conditions at +4 °C. The transfers in which cautious melting was used are indicated in bold. | ||
| Transfer number | Transfer from outdoors | Transfer to forcing |
| 1 | 20 September | 20 September |
| 2 | 4 October | 4 October |
| 3 | 18 October | 18 October |
| 4 | 1 November | 1 November |
| 5 | 15 November | 15 November |
| 6 | 29 November | 6 December |
| 7 | 13 December | 20 December |
| 8 | 3 January | 10 January |
| 9 | 17 January | 24 January |
| 10 | 31 January | 7 February |
| 11 | 28 February | 7 March |
| 12 | 14 March | 21 March |
| 13 | 28 March | 4 April |
| 14 | 11 April | 18 April |
| 15 | 25 April | 25 April |
| 16 | 9 May | 9 May |
The experiment was carried out about twenty years ago. This does not cause any problems for the conclusions of the study because the nursery practices have remained practically unchanged after the time of the experiment.
2.3 Chilling, preforcing, and forcing conditions
The air temperature at the shoot level of the seedlings (30 cm from the ground) was recorded once an hour in the natural outdoor chilling conditions by Tinytalk Data Loggers (Gemini Data Loggers, Chichester, UK) located inside a well-ventilated radiation shield (Fig. 2). The day lengths in the forcing conditions were controlled with black curtains, so that the light period was from 8 am to 4 pm (day length 8 h) and from 4 am to 8 pm (day length 16 h). When necessary, the natural day length was extended by means of metal halide lamps (Philips HPI-T Powertone 400 W Philips, Helmond, the Netherlands). The photosynthetically active radiation at the shoot level was approximately 50 µmol m–2 s–1. The air temperature in the forcing conditions was a constant +20 °C for both day lengths. The seedlings were watered manually with tap water when necessary.
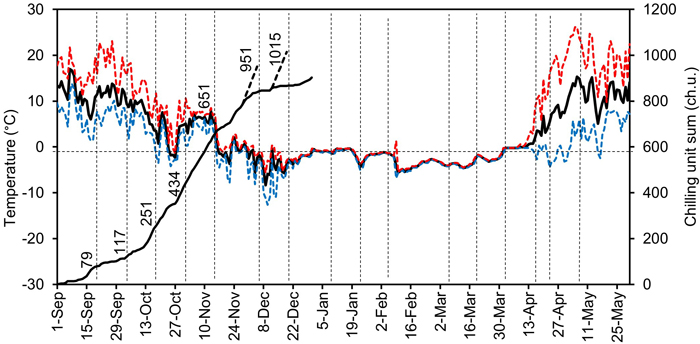
Fig. 2. The daily minimum (blue curve), mean (black curve), and maximum (red curve) outdoor air temperatures from autumn 2005 to spring 2006 (fluctuating curves), and the daily chilling unit sum accumulation (solid black ascending curve on the left) in the experiment carried out at Punkaharju, Finland (61°48´N, 29°20´E). The temperature was measured hourly in the outdoor chilling conditions at the seedling shoot level. The transfer times from the outdoor natural chilling conditions or from the preforcing conditions (+4 °C) applied to melt the seedlings frozen in winter to the forcing conditions in the greenhouse are shown by dashed vertical lines. The figures attached to the curves on the left-hand side of the figure indicate the corresponding chilling unit sum accumulated by each transfer before the end of the year. The chilling unit accumulations in the preforcing conditions are shown by the black dashed ascending curves. The dashed horizontal line indicates the temperature of 0 °C and the chilling unit sum of 600 ch.u.
Whenever the outdoor temperature was below 0°C during the transfers between 29 November and 11 April (Table 1, Fig. 2), the seedlings to be transferred from outdoors to the forcing conditions were first transferred to preforcing conditions (darkness, +4 °C) in cold storage for one week, and only after that were they moved to the forcing conditions in a greenhouse (Fig. 1). This was done to enable cautious thawing and to avoid needle damage (Repo et al. 1984). In the analysis of the data, the day of the transfer from the preforcing to the forcing conditions was taken as the transfer day. The air temperature at the shoot level was recorded once an hour in both the preforcing and the forcing conditions with Tinytalk Data Loggers. In the forcing conditions the loggers were located inside a well-ventilated radiation shield; in the preforcing conditions (in darkness) no shields were applied. On the basis of the hourly temperature measurements, a chilling unit sum was calculated from 1 September to 31 May. The tabulated values of the triangular air temperature response (Sarvas 1974) were used, according to which chilling accumulation (chilling units, ch.u.) takes place in the range of −3.5 to +10.5 °C, with the maximum accumulation rate of 1 chilling unit per hour attained at temperatures between +3.4 and +3.6 °C (Hänninen 2016, p. 74). The minimum, mean, and maximum daily air temperatures and the chilling unit sum accumulated during the autumn are presented in Fig. 2.
2.4 Determination of bud burst
In the forcing conditions, the terminal bud of the main shoot of each seedling was observed visually at an interval of two or three days until bud burst or until the bud was visibly dead as indicated by its dry appearance. The bud was classified as showing bud burst when new needles were visible. For each treatment group, the bud burst percentage, BB%, was calculated as the proportion of seedlings showing bud burst in the terminal bud of the main shoot out of the total number of seedlings observed (n = 10). The days to bud burst, DBB, was calculated as the mean (or the median; see below) number of days from the beginning of forcing to the day of the observed terminal bud burst. In addition, the bud burst of the terminal bud of the main shoot of twenty SD-treated and twenty control seedlings was observed in natural outdoor conditions in May and early June.
2.5 Statistical analyses
The differences of the bud burst percentage, BB%, between treatments were analysed by means of a logistic regression with binary response (bud burst / no bud burst) of the terminal bud:
![]()
where p is the probability of bud burst and m is the intercept. S is the combined photoperiodic effect (short day vs. control outdoors – 8 h vs. 16 h day length in indoors forcing), consisting of four levels: 1) SD outdoors and 8 h forcing, 2) control outdoors and 8 h forcing, 3) SD outdoors and 16 h forcing, and 4) control outdoors and 16 h forcing). T is the transfer time to forcing with 16 levels, and ST is the interaction of S and T. The differences of the number of days to bud burst, DBB, between the treatments were analysed with a two-way analyses of variance:
![]()
where e is the error term, other symbols as in Eq. (1). The experimental unit in both of the analyses was the seedling and the number of seedling replicates was n = 10. In both of the analyses, the interaction term was found highly significant. At each transfer time the differences between the photoperiodic treatments were tested pairwise with Fisher’s exact test (BB%) and the LSD test (DBB). Statistical analyses were carried out with IBM SPSS Statistics for Winfows, Version 26.0., IBM Corp., Armonk, NY, USA.
3 Results
Both the SD treatment in the previous summer and the long photoperiod in the forcing conditions reduced the chilling requirement in Norway spruce seedlings (see Fig. 1 for the experimental design, Fig. 3, Supplementary file S1). The effects of the photoperiodic treatments and the transfer time on BB% and their interaction were all significant (Suppl. file S2). In forcing with both the long photoperiod (16 h) and the short photoperiod (8 h), BB% increased from zero towards 100% with increasing previous chilling accumulation earlier in the SD-treated seedlings than in the control seedlings. Similarly, the increase occurred earlier in the long photoperiod than in the short photoperiod forcing. In both cases, the difference was about 14 days, corresponding approximately to 200 ch.u. in the chilling requirement. In the third transfer (18 October), BB% was significantly higher in the SD-treated seedlings forced in the long photoperiod than in seedlings that had undergone any of the other three treatments (Suppl. file S1). In the fourth transfer (1 November), BB% was significantly lower in the control seedlings forced in the short photoperiod than in the seedlings that had undergone any of the other three treatments. From 6 December (the sixth transfer to forcing, 951 ch.u.) on, BB% stayed at 100% in all transfers, with two exceptions until the end of the experiment in early May (Suppl. file S1).
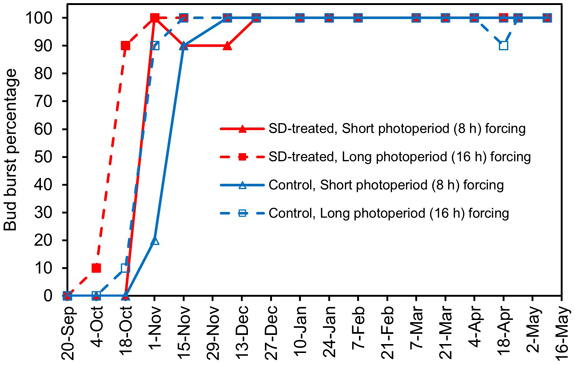
Fig. 3. Bud burst percentage, BB%, of the terminal buds in second-year SD-treated seedlings (SD, red lines and solid red symbols) and control seedlings (blue lines and empty blue symbols) of Norway spruce in a chilling-forcing experiment carried out with day lengths of 8 h (triangles, solid lines) and 16 h (squares, dashed lines) in the forcing conditions. The SD treatment was performed in late summer 2005, and after that, the seedlings were chilled in outdoor natural conditions. The horizontal axis indicates the dates when the seedlings were transferred from outdoor natural chilling conditions (or from the preforcing conditions at +4 °C) to a regrowth test under forcing conditions in a greenhouse (see Table 1, Figs. 1 and 2). The standard error of the mean BB% varied between 0.0 and 12.6%.
SD treatment in previous summer reduced DBB in both long and short photoperiod forcing (Fig. 4, Suppl. files S3 and S4). The effects of the photoperiodic treatment and the transfer time on DBB and their interaction were all significant (Suppl. file S5). In all four treatments from the fourth transfer (1 November) on, DBB generally decreased in the forcing conditions with successively later transfers and increasing previous chilling accumulation (Fig. 4, Suppl. file S3). In some transfers, however, the value of DBB transiently increased. When the median was used instead of the mean, the temporary increase diminished in the DBB curves (Fig. 4, Suppl. file S3). Until the ninth transfer (24 January), in forcing with both the long and the short photoperiod, the buds of the SD-treated seedlings burst earlier than the buds of the control seedlings (Fig. 4; Suppl. files S3 and S4). The buds of the control seedlings burst earlier in long-day than in short-day forcing in transfers until the eighth transfer on 10 January. In contrast, the buds of the SD-treated seedlings burst at almost the same time in short-day and long-day forcing as early as before 24 January (Fig. 4, Suppl. files S3 and S4). In the transfers between 7 February and 21 March, the DBB curves decreased gently or even rose in some transfers. In the transfers of April and May, the DBB curves decreased steeply (Fig. 4, Suppl. file S3).
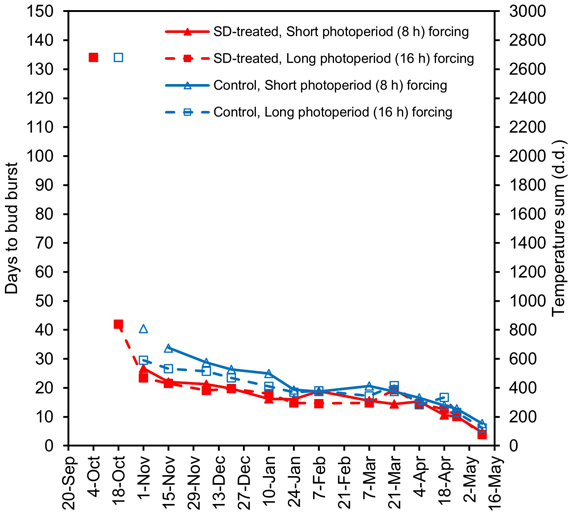
Fig. 4. Mean days to bud burst, DBB, from the beginning of forcing in second-year SD-treated seedlings (SD, red lines and solid red symbols) and control seedlings (blue lines and empty blue symbols) of Norway spruce in a chilling-forcing experiment carried out with day lengths of 8 h (triangles, solid lines) and 16 h (squares, dashed lines) in the forcing conditions. The SD treatment was performed in late summer 2005, and after that, the seedlings were chilled in outdoor natural conditions. The horizontal axis indicates the dates when the seedlings were transferred from outdoor natural chilling conditions (or from the preforcing conditions at +4 °C) to a regrowth test under forcing conditions in a greenhouse (Table 1, Figs. 1 and 2). The vertical axis on the right-hand side of the figure shows the approximate temperature sum with the threshold temperature of 0 °C in forcing conditions corresponding to the DBB. The standard error of the mean DBB varied between 1.5 and 4.8 days. The data points not connected with the corresponding line indicate observations based on low numbers (1–2) of seedlings showing bud burst.
In natural outdoor conditions, the SD treatment of the previous summer advanced bud burst until 24 May (Fig. 5). Eleven of the observed twenty SD-treated seedlings (55%) showed bud burst on 15 May, whereas among the control seedlings, approximately the same percentage (50%) was obtained one week later, on 22 May. From 26 May on, there was no difference any longer in the bud burst percentage between the SD-treated and the control seedlings (Fig. 5).
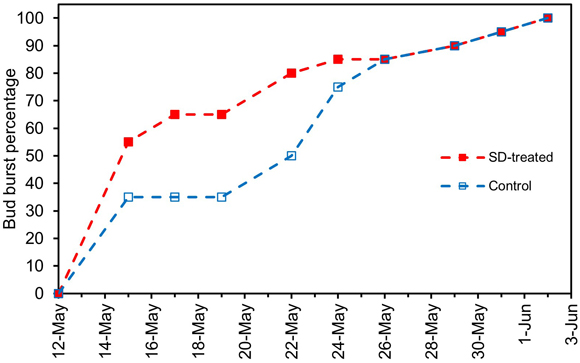
Fig. 5. Development of the bud burst percentage, BB%, of the terminal buds in SD-treated seedlings (SD, red lines and solid red symbols) and control seedlings (blue lines and empty blue symbols) of Norway spruce under natural outdoor conditions in spring 2006. The SD treatment was performed in late summer 2005.
4 Discussion
In the present study, late-summer SD treatment reduced the chilling requirement of the Norway spruce seedlings in the next autumn. Consequently, the second-year Norway spruce seedlings that were subjected to an artificial SD treatment in the preceding summer attained full ontogenetic competence in the autumn with less chilling accumulation than the control seedlings did, showing that endodormancy released earlier in the SD-treated than the control seedlings. This difference was seen in the forcing conditions until late January. In other words, the SD treatment hastened the development of the seedlings during dormancy. A possible reason for that could be that SD treatment advanced the growth cessation, bud set, and endodormancy induction in late summer and early autumn, thus giving the SD-treated seedlings a lead in their developmental stage in comparison with the control seedlings. Moreover, long-photoperiod (16 h) forcing advanced the increase of BB% in the seedlings during autumn (Fig. 3). This suggests that long-day forcing partially compensated for the lack of chilling, which is in line with earlier studies of Norway spruce (Nienstaedt 1967; Worrall and Mergen 1967). In the present study, the short-photoperiod (8 h) forcing delayed the increase of BB% in the seedlings during the autumn before the chilling requirement was fulfilled (Fig. 3). This is in line with Basler and Körner (2012), who found that photoperiods shorter than 12 h delay endodormancy release and bud burst in Norway spruce.
The DBB curves of the control seedlings declined steeply in the transfers until 24 January (Fig. 4, Suppl. file S3). This suggests that the prolonged chilling increased the rate of internal microscopic development of the bud (Hänninen 2016 and references therein), thus shortening the DBB of the control seedlings to nearly the same level as in the SD-treated seedlings. Until 24 January, the SD-treated seedlings showed bud burst earlier than the control seedlings in both long-photoperiod and short-photoperiod forcing. In the transfers between 7 February and 21 March, the DBB curves declined gently and even rose in some transfers. The rise was caused by some outlying seedlings. For some reason their bud burst happened much later than that of the other seedlings. In these cases the use of the median instead of the mean reduced this unexpected deviation from the generally decreasing trend of DBB (Fig. 4, Suppl. file S3). The levelling off of the DBB curves suggests that the seedlings had achieved full ontogenetic competence (Hänninen 1990, 1995). In the transfers of April and May, the DBB curves declined very steeply, suggesting that the internal development of the buds was already going on in natural conditions before the seedlings were moved to the forcing conditions (Sutinen et al. 2012).
From early autumn till late January, the DBB of the control seedlings was shorter in long- photoperiod than in short-photoperiod forcing. The reason for this could be that less chilling is needed for bud burst in long photoperiod than in short photoperiod. The buds of many Picea species, including Picea abies, can burst in a 16–20 h photoperiod (long days) at normal growth temperatures even without previous chilling (Nienstaedt 1967; Worrall and Mergen 1967). The DBB of the SD-treated seedlings was shorter than that of the control seedlings, and it was almost similar in both photoperiods (Fig. 4, Suppl. file S3). This suggests that photoperiod has no effect on the time to bud burst in SD-treated seedlings, even though it did affect the occurrence of bud burst, as indicated by the bud burst percentage. In other words, the role of forcing photoperiod in the bud burst of the SD-treated seedlings seemed to be less important than in the bud burst of the control seedlings. Consequently, under natural conditions the buds of the SD-treated seedlings were able to burst in lower photoperiod and thus earlier than those of the control seedlings. Photoperiod remains the same from year to year in each location and is therefore not dependent on the climate change and variation in weather conditions.
In natural outdoor conditions, the SD treatment advanced bud burst slightly (Fig. 5). The possible reasons for this could be that as an SD treatment advances the development of the seedlings, they are quicker to attain the developmental phase where photoperiod no longer regulates their bud burst, which makes the buds of the SD-treated seedlings able to burst earlier than the control seedlings in natural conditions (i.e., with shorter photoperiod). The result of SD treatment advancing bud burst is in line with earlier studies (Heide 1974b; Sandvik 1980; Fløistad and Granhus 2010). It is also in line with Luoranen and Sutinen (2017). They found that the buds of SD-treated seedlings had fewer and shorter protective bud scales than control seedlings and suggested that these changes in bud structure may partly explain the early bud burst of SD- treated seedlings. On the other hand, some earlier studies have shown that relatively high temperatures during SD exposure increase the depth of dormancy in many tree species, including Norway spruce, thus increasing the high-temperature requirement for bud burst and leading to later bud burst in the next spring (Dormling 1989; Heide 2003; Søgaard et al. 2008; Kalcsits et al. 2009; Malyshev 2020; Beil et al. 2021; Garrigues et al. 2023). This quantitative dormancy induction (Hänninen 2016) did not, however, occur in the present study. One possible reason for this could be that the night temperatures during the SD exposure were lower, which modified the bud-burst-delaying effect of higher day temperatures on bud formation in a complex way (Olsen et al. 2014). According to Olsen et al. (2014), the fastest bud burst occurs at intermediate night temperatures during SD exposure, and bud burst is delayed by both lower and higher night temperatures.
5 Conclusions
The main finding of the present study is that SD treatment during the preceding summer reduced the chilling requirement and advanced the endodormancy release in Norway spruce seedlings in the autumn. In addition, long photoperiod in forcing conditions reduced the chilling requirement. In the autumn, up to late January, the terminal buds of the SD-treated seedlings burst earlier and generally at almost the same time in both photoperiods, but those of the control seedlings burst later and in a shorter time in the long photoperiod than in the short one. This suggests that the photoperiod prevailing in forcing conditions has no additional delaying or promoting effect on the bud burst of SD-treated seedlings. In natural outdoor conditions, the SD treatment advanced bud burst slightly. The buds of SD-treated seedlings are able to burst earlier and in shorter photoperiod than those of control seedlings. Thus, the growing season of SD-treated seedlings can start earlier and be slightly longer. This favours both nurseries and forest management. However, further studies are needed on the interaction of SD treatment and photoperiod.
Declaration of the use of generative artificial intelligence and AI-assisted technologies
No generative artificial intelligence or AI-assisted technologies were used in the writing process.
Declaration of openness of research materials, data, and code
The data are available through the open research repository Figshare: https://doi.org/10.6084/m9.figshare.31998786. No computer code was used in the study.
Author contributions
JP: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Validation, Visualization, Writing – original draft.
RH: Conceptualization, Formal analysis, Methodology, Visualization, Writing – original draft
A-VA: Writing – original draft.
NS: Writing – original draft.
HH: Conceptualization, Methodology, Visualization, Writing – original draft.
Acknowledgments
We wish to thank Jouko Lehto for assistance in running the greenhouse experiments and Jaakko Heinonen for statistical advice.
Funding
This research was supported by the Finnish Forest Research Institute (currently part of the Natural Resources Institute Finland, Luke) (projects 3365 and 3538). Also, the Emil Aaltonen Foundation, the Foundation for Forest Tree Breeding, the Niemi Foundation (grants No. 20190006, 20200015, 20210030, 20220033), and the Finnish Cultural Foundation, the South Savo Regional fund (grants No. 12191814, 12231922) provided research grants to JP.
References
Augspurger CK (2013) Reconstructing patterns of temperature, phenology, and frost damage over 124 years: spring damage risk is increasing. Ecology 94: 41–50. https://doi.org/10.1890/12-0200.1.
Basler D, Körner C (2012) Photoperiod sensitivity of bud burst in 14 temperate forest tree species. Agric For Meteorol 165: 73–81. https://doi.org/10.1016/j.agrformet.2012.06.001.
Beil I, Kreyling J, Meyer C, Lemcke N, Malyshev AV (2021) Late to bed, late to rise—warmer autumn temperatures delay spring phenology by delaying dormancy. Glob Change Biol 27: 5806–5817. https://doi.org/10.1111/gcb.15858.
Colombo SJ Menzies MI, O’Reilly C (2001) Influence of nursery cultural practices on cold hardiness of coniferous forest tree seedlings. In: Bigras FJ, Colombo SJ (eds) Conifer cold hardiness. Kluwer Academic Publishers, Dordrecht, The Netherlands, pp 223–252. https://doi.org/10.1007/978-94-015-9650-3_9.
Dormling I (1989) The role of photoperiod and temperature in in the induction and the release of dormancy in Pinus sylvestris L. seedlings. Ann For Sci 46: 228–232. https://doi.org/10.1051/forest:19890554.
Dormling I, Gustafsson, A, von Wettstein D (1968) The experimental control of the life cycle in Picea abies (L.) Karst. I. Some basic experiments on the vegetative cycle. Silvae Genet 17: 44–64.
Ekberg I, Eriksson G, Dormling I (1979) Photoperiodic reactions in conifer species. Holarctic Ecol 2: 255–263. https://doi.org/10.1111/j.1600-0587.1979.tb01297.x.
Fløistad IS, Granhus A (2010) Bud break and spring frost hardiness in Picea abies seedlings in response to photoperiod and temperature treatments. Can J For Res 40: 968–976. https://doi.org/10.1139/X10-050.
Fløistad IS, Granhus A (2019) Morphology and phenology in Picea abies seedlings in response to split short day treatments. Baltic For 25: 38–44. https://doi.org/10.46490/vol25iss1pp038.
Fuchigami LH, Weiser CJ, Kobayashi K, Timmis R, Gusta LV (1982) A degree growth stage (°GS) model and cold acclimation in temperate woody plants. In: Li PH, Sakai A (eds) Plant cold hardiness and freezing stress. Mechanisms and crop implications. Academic Press, New York, pp 93–116. https://doi.org/10.1016/B978-0-12-447602-8.50012-X.
Garrigues R, Dox I, Flores O, Marchand LJ, Malyshev AV, Beemster G, AbdElgawad H, Janssens I, Asard H, Campioli M (2023) Late autumn warming can both delay and advance spring budburst through contrasting effects on bud dormancy depth in Fagus sylvatica L.. Tree Physiol 43: 1718–1730. https://doi.org/10.1093/treephys/tpad080.
Hänninen H (1990) Modelling bud dormancy release in trees from cool and temperate regions. Acta For Fenn 213: 1–47. https://doi.org/10.14214/aff.7660.
Hänninen H (1995) Effects of climatic change on trees from cool and temperate regions: an ecophysiological approach to modelling of bud burst phenology. Can J Bot 73: 183–199. https://doi.org/10.1139/b95-022.
Hänninen H (2016) Boreal and temperate trees in a changing climate: modelling the ecophysiology of seasonality. Biometeorology 3. Springer, Dordrecht. https://doi.org/10.1007/978-94-017-7549-6.
Heide OM (1974a) Growth and dormancy in Norway spruce ecotypes (Picea abies) I. Interaction of photoperiod and temperature. Physiol Plant 30: 1–12. https://doi.org/10.1111/j.1399-3054.1974.tb04983.x.
Heide OM (1974b) Growth and dormancy in Norway spruce ecotypes II. After-effects of photoperiod and temperature on growth and development in subsequent years. Physiol Plant 31: 131–139. https://doi.org/10.1111/j.1399-3054.1974.tb03117.x.
Heide OM (2003) High autumn temperature delays spring bud burst in boreal trees, counterbalancing the effect of climatic warming. Tree Physiol 23: 931–936. https://doi.org/ 10.1093/treephys/23.13.931.
Junttila O (2007) Regulation of annual shoot growth cycle in northern tree species. In: Taulavuori E, Taulavuori K (eds) Physiology of northern plants under changing environment. Research Signpost, Kerala, India, pp 177–210.
Juntunen M-L, Rikala R (2001) Fertilization practice in Finnish forest nurseries from the standpoint of environmental impact. New For 21: 141–158. https://doi.org/10.1023/A:1011837800185.
Kalcsits LA, Silim S, Tanino K (2009) Warm temperature accelerates short-photoperiod-induced growth cessation and dormancy induction in hybrid poplar (Populus × spp.). Trees 23: 971–979. https://doi.org/10.1007/s00468-009-0339-7.
Koski V, Selkäinaho J (1982) Experiments on the joint effect of heat sum and photoperiod on seedlings of Betula pendula. Commun Inst For Fenn 105: 1–34. https://urn.fi/URN:ISBN:951-40-0574-0.
Koski V, Sievänen R (1985) Timing of growth cessation in relation to the variations in the growing season. In: Tigerstedt PMA, Puttonen P, Koski V (eds) Crop physiology of forest trees. Helsinki University Press, Helsinki, pp 167–193.
Konttinen K, Rikala R, Luoranen J (2003) Timing and duration of short-day treatment of Picea abies seedlings. Baltic For 9: 2–9.
Lang GA, Early JD, Martin, GC, Darnell RL (1987) Endo-, para-, and ecodormancy: physiological terminology and classification for dormancy research. HortSci 22: 371–377. https://doi.org/10.21273/HORTSCI.22.3.371.
Lilja A, Poteri M, Petäistö R-L, Rikala R, Kurkela T, Kasanen R (2010) Fungal diseases in forest nurseries in Finland. Silva Fenn 44: 525–545. https://doi.org/10.14214/sf.147.
Luoranen J, Sutinen S (2017) Reduced height of short day induced bud scale complex may partly explain early bud burst in Norway spruce seedlings. Silva Fenn 51, article id 7759. https://doi.org/10.14214/sf.7759.
Ma Q, Huang, J-G, Hänninen H, Berninger F (2019) Divergent trends in the risk of spring frost damage to trees in Europe with recent warming. Glob Chang Biol 25: 351–360. https://doi.org/10.1111/gcb.14479.
Malyshev AV (2020) Warming events advance or delay spring phenology by affecting bud dormancy depth in trees. Front Plant Sci 11, article id 856. https://doi.org/10.3389/fpls.2020.00856.
Nienstaedt H (1967) Chilling requirements in seven Picea species. Silvae Genet 16: 65–68.
Nikkanen T, Karvinen K, Koski V, Rusanen M, Yrjänä-Ketola L (1999) Kuusen ja männyn siemenviljelykset ja niiden käyttöalueet. [Seed orchards of Norway spruce and Scots pine and areas of usage of their seeds]. Metsäntutkimuslaitoksen tiedonantoja 730: 1–203. https://urn.fi/URN:ISBN:951-40-1677-7.
Olsen JE, Lee Y, Junttila O (2014) Effect of alternating day and night temperature on short day induced bud set and subsequent bud burst in long days in Norway spruce. Front Plant Sci 5, article id 691. https://doi.org/10.3389/fpls.2014.00691.
Partanen J, Beuker E (1999) Effects of photoperiod and thermal time on the growth rhythm of Pinus sylvestris seedlings. Scand J For Res 14: 487–497. https://doi.org/10.1080/02827589908540813.
Partanen J, Koski V, Hänninen H (1998) Effects of photoperiod and temperature on the timing of bud burst in Norway spruce (Picea abies). Tree Physiol 18: 811–816. https://doi.org/10.1093/treephys/18.12.811.
Partanen J, Leinonen I, Repo T (2001) Effect of accumulated duration of the light period on bud burst in Norway spruce (Picea abies) of varying ages. Silva Fenn 35: 111–117. https://doi.org/10.14214/sf.608.
Partanen J, Häkkinen R, Sutinen S, Viherä-Aarnio A, Zhang R, Hänninen H (2021) Endodormancy release in Norway spruce grafts representing trees of different ages. Tree Physiol 41: 631–643. https://doi.org/10.1093/treephys/tpaa001.
Repo T, Mela M, Valtanen J (1984) Männyn versosyövälle alttiiden ja vastustuskykyisten taimialkuperien erottaminen neulasten ominaisuusimpedanssin mittauksella. [Separation of susceptible and resistant provenances of Scots pine to Gremeniella abietina by specific needle impedance]. Folia For 610: 1–11. https://urn.fi/URN:ISBN:951-40-0684-4.
Riikonen J, Luoranen J (2020) An assessment of storability of Norway spruce container seedlings in freezer storage as affected by short-day treatment. Forests 11, article id 692. https://doi.org/10.3390/f11060692.
Riikonen J, Ruhanen H, Luoranen J (2023) Impact of warm spells during late fall and winter on frost hardiness of short-day treated Norway spruce seedlings. For Ecol Manage 542, article id 121105. https://doi.org/10.1016/j.foreco.2023.121105.
Romberger JA (1963) Meristems, growth, and development in woody plants. An analytical review of anatomical, physiological, and morphogenetic aspects. USDA Technical Bulletin 1292. U.S. Government Printing Office, Washington DC.
Ruosteenoja K, Markkanen T, Räisänen J (2020) Thermal seasons in northern Europe in projected future climate. Int J Climatol 40: 4444–4462. https://doi.org/10.1002/joc.6466.
Sandvik M (1980) Environmental control of winter stress tolerance and growth potential in seedlings of Picea abies (L.) Karst. N Z J For Sci 10: 97–104.
Sarvas R (1974) Investigations on the annual cycle of development of forest trees II. Autumn dormancy and winter dormancy. Commun Inst For Fenn 84: 1–101. https://urn.fi/URN:NBN:fi-metla-201207171117.
Søgaard G, Johnsen Ø, Nilsen J, Junttila O (2008) Climatic control of bud burst in young seedlings of nine provenances of Norway spruce. Tree Physiol 28: 311–320. https://doi.org/10.1093/treephys/28.2.311.
Sutinen S, Partanen J, Viherä-Aarnio A, Häkkinen R (2012) Development and growth of primordial shoots in Norway spruce before visible bud burst in relation to time and temperature in the field. Tree Physiol 32: 987–997. https://doi.org/10.1093/treephys/tps063.
Svystun T, Lundströmer J, Berlin M, Westin J, Jönsson, AM (2021) Model analysis of temperature impact on the Norway spruce provenance specific bud burst and associated risk of frost damage. For Ecol Manage 493, article id 119252. https://doi.org/10.1016/j.foreco.2021.119252.
Tanino KK, Kalcsits L, Silim S, Kendall E, Gray GR (2010) Temperature-driven plasticity in growth cessation and dormancy development in deciduous woody plants: a working hypothesis suggesting how molecular and cellular function is affected by temperature during dormancy induction. Plant Molec Biol 73: 49–65. https://doi.org/10.1007/s11103-010-9610-y.
Vaartaja O (1959) Evidence of photoperiodic ecotypes in trees. Ecol Monogr 29: 91–111. https://doi.org/10.2307/1942199.
Viherä-Aarnio A, Sutinen S, Partanen J, Häkkinen R (2014) Internal development of vegetative buds of Norway spruce trees in relation to accumulated chilling and forcing temperatures. Tree Physiol 34: 547–556. https://doi.org/10.1093/treephys/tpu038.
Wallin E, Gräns D, Jacobs DF, Lindström A, Verhoef N (2017) Short-day photoperiods affect expression of genes related to dormancy and freezing tolerance in Norway spruce seedlings. Ann For Sci 74, article id 59. https://doi.org/ 10.1007/s13595-017-0655-9.
Wareing PF (1956) Photoperiodism in woody plants. Annu Rev Plant Physiol 7: 191–214. https://doi.org/10.1146/annurev.pp.07.060156.001203.
Worrall J, Mergen F (1967) Environmental and genetic control of dormancy in Picea abies. Physiol Plant 20: 733–745. https://doi.org/10.1111/j.1399-3054.1967.tb07217.x.
Total of 51 references.

