Effects of selection harvesting on the understorey vegetation in drained Norway spruce peatlands
Hotanen J.-P., Miina J., Korpela L., Mäkipää R. (2026). Effects of selection harvesting on the understorey vegetation in drained Norway spruce peatlands. Silva Fennica vol. 60 no. 1 article id 25061. https://doi.org/10.14214/sf.25061
Highlights
- The thinning intensity and time since harvest significantly affect vegetation dynamics on fertile, drained Norway spruce peatlands
- Several species respond to the thinning intensity, showing a greater treatment effect with more time since thinning
- Vaccinium myrtillus increases, especially on the most intensively thinned plots
- Selection harvesting has a minor effect on the number of plant species in drained Norway spruce peatlands.
Abstract
This study focused on the changes in species abundance after harvest in uneven-aged stands. Selection harvesting was performed at four sites in southern boreal vegetation zones in Finland using two thinning intensities: post-harvest basal area (G) of 17 and 12–13 m2 ha–1. The G pre-harvest and in control plots varied between 19 and 31 m2 ha–1. Vegetation was inventoried before thinning in 2016 and 2 and 6 years after thinning in 2018 and 2022. The effect of thinning intensity was significant for grasses and sedges as groups, Betula pubescens Ehrh. (height < 50 cm), Rubus idaeus L., and Trientalis europaea L., which showed increased abundance after harvest. However, T. europaea abundance turned to decline by 2022. Several species responded to the thinning intensity, showing a greater treatment effect with the more time since harvest. The abundance of Carex globularis L., Dryopteris carthusiana (Vill.) H.P. Fuchs, Epilobium angustifolium L., Vaccinium myrtillus L., V. vitis-idaea L., Linnaea borealis L. and Brachythecium spp. increased, but that of Oxalis acetosella L. decreased. For some species, only the time since harvest was significant. The abundance of Maianthemum bifolium (L.) F.W. Schmidt, Deschampsia flexuosa (L.) Trin. and Plagiothecium spp. increased, whereas that of Sphagnum girgensohnii Russow and S. russowii Warnst. decreased. The thinning intensity did not have a significant effect on the number of species, but the number of species increased slightly on the thinned plots. The effects of logging residues, strip roads, and light availability may be the major drivers of the changes in the species abundance.
Keywords
continuous cover forestry;
ground vegetation;
thinning from above;
ANOVA;
uneven-aged cutting
- Hotanen, Natural Resources Institute Finland (Luke), Yliopistokatu 6 B, FI-80100 Joensuu, Finland E-mail ext.juha-pekka.hotanen@luke.fi
-
Miina,
Natural Resources Institute Finland (Luke), Yliopistokatu 6 B, FI-80100 Joensuu, Finland
 https://orcid.org/0000-0002-8639-4383
E-mail
jari.miina@luke.fi
https://orcid.org/0000-0002-8639-4383
E-mail
jari.miina@luke.fi
-
Korpela,
Natural Resources Institute Finland (Luke), Latokartanonkaari 9, FI-00790 Helsinki, Finland
 https://orcid.org/0000-0001-9900-4332
E-mail
ext.leila.korpela@luke.fi
https://orcid.org/0000-0001-9900-4332
E-mail
ext.leila.korpela@luke.fi
-
Mäkipää,
Natural Resources Institute Finland (Luke), Latokartanonkaari 9, FI-00790 Helsinki, Finland
 https://orcid.org/0000-0003-3146-4425
E-mail
raisa.makipaa@luke.fi
https://orcid.org/0000-0003-3146-4425
E-mail
raisa.makipaa@luke.fi

Received 20 November 2025 Accepted 15 March 2026 Published 27 March 2026
Views 1724
Available at https://doi.org/10.14214/sf.25061 | Download PDF

Supplementary Files
1 Introduction
In a boreal forest ecosystem, understorey vegetation plays a significant role in forest regeneration, the nutrient cycle, and biomass production (Nilsson and Wardle 2005). Understorey vegetation and trees determine the amount and quality of plant litter and regulate soil nutrient cycles and balance in addition to microbial communities and environmental factors, such as temperature, relative humidity, sunlight and soil aeration. Silviculture has a significant impact on the understorey in peatland and upland mineral soils (Moilanen et al. 1995; Saarinen 2013; Tarvainen et al. 2015; Tonteri et al. 2016; Vanha-Majamaa et al. 2017; Hamberg et al. 2019). Harvesting changes the growth conditions by increasing the amount of sunlight and causing variations in the air and soil temperatures. It also affects humidity and nutrient mobilisation in the soil (Hannertz and Hånell 1997; Rydgren et al. 2004; Heithecker and Halpern 2007; Päivänen and Hånell 2012; Sarkkola et al. 2012; Leppä et al. 2020b).
In Finland, most forest regeneration takes place by clearcutting (Korhonen et al. 2024). In peatland forests, stand regeneration often includes ditch network maintenance after clearcutting. The strong impact of clearcutting on biodiversity, waterbodies, greenhouse gas (GHG) emissions and landscapes has attracted interest in alternative silvicultural methods. The 2014 reform of the Forest Act enabled more diverse forest management practices; thus, selection harvesting, which enables continuous cover forestry, was permitted (Äijälä et al. 2014).
In continuous cover forestry, the intensity of harvesting affects the quantity and structure of the remaining stand. In selection harvesting, trees of all size classes are removed, focusing on the removal of larger trees, similar to thinning from above, while always leaving enough larger trees to ensure natural seed production and forest regeneration (Saarinen et al. 2020). Continuous cover forestry is seen as an alternative to rotation forestry, especially in peatlands (Nieminen et al. 2018; Saarinen et al. 2020). As peatlands account for roughly one-fourth of Finland’s entire forest land area and the growing stock volume (Korhonen et al. 2024), they comprise a significant portion of the future harvesting potential.
The impact of understorey vegetation succession on natural regeneration must be understood (Saarinen 2013). Seedling emergence in small gaps in Norway spruce peatlands in Northern Finland is particularly high (Hökkä and Mäkelä 2014; Hökkä and Repola 2018). Natural regeneration in spruce-dominated peatland forests based on shelterwood harvesting has produced promising results (Holgén and Hånell 2000). However, understorey vegetation may quickly overgrow regeneration areas in Norway spruce peatlands, and preparation tracks may be covered with moss after clearcutting, decelerating the initial development of natural and planted seedlings (Moilanen et al. 1995, 2011; Hannertz and Hånell 1997).
The dynamics of understorey vegetation following selection harvesting have not been widely studied on drained peatlands. The impact of different harvesting methods, including those belonging to continuous cover forestry, on the undergrowth structure has been studied in mineral soils (Vanha-Majamaa et al. 2017 and references therein). Studies in peatlands have mainly focused on the first stages of vegetation succession after clearcutting (Moilanen et al. 1995; Saarinen et al. 2009; Hamberg et al. 2019). Haapakoski et al. (2021) studied the short-term effects (two-year period) of selection harvesting on the structure of understorey vegetation. In addition, the relationship of bilberry (Vaccinium myrtillus L.) and lingonberry (Vaccinium vitis-idaea L.) coverage and berry yield with peatland tree stand characteristics were studied by Turtiainen et al. (2013, 2016). Considering biomass production, evaporation and competition in the entire community, including tree seedlings, these long-lived dwarf shrubs that spread through underground stems are a key part of the understorey.
This study examined changes in the abundance of plant species and species groups as well as species richness caused different intensities of selection harvesting. Because selection harvesting partially opens the canopy, we expect moderate changes in understorey cover and composition compared to unharvested controls. We hypothesised that harvesting intensity, time since harvest, and their interaction affects vegetation dynamics on fertile drained peatlands.
2 Material and methods
2.1 Sample plots and vegetation data
The effect of selection harvesting on understory vegetation was experimentally studied at four drained peatland sites in the southern boreal zones of Janakkala, Multia, Heinävesi, and Juuka municipalities in southern Finland (Leppä et al. 2020a). In addition to no cutting, two thinning intensities were tested at the study sites, resulting in remaining stand basal areas of 12–13 m2 ha–1 with heavy cutting and 17 m2 ha–1 with moderate cutting (Table 1). In spruce-dominated stands with a J-shape diameter distribution (Leppä et al. 2020a), the experimental plots (40 m × 40 m, located within the sites) were thinned to the target basal area so that the remaining stand included trees of various sizes and enough dominant trees to ensure seed production and forest regeneration. However, harvest removal was emphasised on the upper half of the stand diameter at breast height (DBH) distribution. The forest types of the experimental sites were herb-rich and Vaccinium myrtillus drained peatland (Laine et al. 2018) (Table 1).
| Table 1. Information on the sample plots. Remaining stand basal area (Gafter) in moderate (less intensive) and heavy (intensive) cutting was 17 and 12–13 m2 ha–1, respectively. Gbefore = stand basal area before cutting. n = number of sample plots. Drained peatland forest type (Laine et al. 2018), Rhtkg (I) and (II) = herb-rich types (I) and (II), Mtkg(I) = Vaccinium myrtillus type (I), Mtkg(I-) = more infertile border variant of Mtkg(I). | |||||
| Municipality | Treatment | Gafter | Gbefore | n | Site type |
| Heinävesi (Rouvanlehto) | Control | 22–23 | 22–23 | 2 | Rhtkg(I) |
| Moderate | 17 | 21–24 | 2 | Rhtkg(I) | |
| Heavy | 12 | 22 | 2 | Rhtkg(I) | |
| Janakkala (Paroninkorpi) | Control | 22–30 | 22–30 | 5 | Rhtkg(II) |
| Moderate | 17 | 22–27 | 5 | Rhtkg(II) | |
| Heavy | 12 | 23–31 | 5 | Rhtkg(II) | |
| Juuka (Vaarajoki) | Control | 20–22 | 20–22 | 2 | Mtkg(I) |
| Moderate | 17 | 19–24 | 2 | Mtkg(I) | |
| Heavy | 12 | 25–29 | 2 | Mtkg(I) | |
| Multia (Havusuo) | Control | 25–28 | 25–28 | 2 | Mtkg(I) – Mtkg(I-) |
| Heavy | 13 | 29–31 | 2 | Mtkg(I) – (Mtkg(I-) | |
Vegetation was inventoried on each experimental plot from 15 systematically placed 1 m2 vegetation inventory squares immediately before cutting in 2016 and again 2 years (2018) and 6 years after cutting (2022). The squares were located on 3 parallel lines (5 per line) on the plot. Their exact location was marked on the overlay drawing of the plot and with sticks on the ground. The projection coverage of field- and ground-layer species was assessed on a scale of 0.1, 0.2, 0.3, 0.5, 1, 2, 3, 5, 7, 10, 15, …, 95, 97, 98, … 100%. Rubus idaeus L. and Salix aurita L. were assessed without a height limit, but for trees, a maximum limit of 0.5 m was used. The coverage of forest litter, which was mainly composed of dead needles, leaves, and small woody debris, was also assessed. The scientific names of the species followed the nomenclature described by Hämet-Ahti et al. (1998) for vascular plants and Ulvinen et al. (2002) for mosses (which is in concordance with Missouri Botanical Garden nomenclature, https://www.tropicos.org).
The sampling hierarchy included 4 drained peatland sites, 4–15 experimental plots per site, and 15 vegetation squares per plot. For data analysis, species cover on the 15 vegetation inventory squares were averaged for each experimental plot.
2.2 Statistical analyses
Due to the skewed distributions of the response variables, the logarithmic transformation of the percent cover was used to avoid heteroscedasticity, reduce skewness (McDonald 2014) and linearise multiplicative relationships. Log transformation of data with zero values required adding a small constant (1). Bounded back-transformed predictions (0–100) were obtained because they are never negative, and the observed (and predicted) coverages of plant species groups and species were less than 100% (Supplementary file S1).
The data had spatially hierarchical (experimental plots within peatland sites) and temporal (measurement occasions) correlation structures. Correlated observations were considered by dividing residual variation into between-site, between-plot, and within-plot components. The within-plot error terms were assumed to arise from a first-order autoregressive process.
Two-way repeated measures analysis of variance (ANOVA) was used to determine how the percent cover of the species or species group was affected by the harvesting intensity and time since treatment:
![]()
where the dependent variable is the ln-transformed percent cover of the species or species group on treatment plot i of the site j (j = 1, 2, 3, 4) in year t; μ is the overall mean; Treatment is the harvesting intensity applied to the treatment plot (k = no cutting, moderate cutting, heavy cutting); Year is the measurement year (t = 2016, 2018, 2022); uj is the random site effect ![]() uij is the random treatment plot effect
uij is the random treatment plot effect ![]()
![]() is the autocorrelated error term of treatment plot i of site j in year t,
is the autocorrelated error term of treatment plot i of site j in year t, ![]() and
and ![]()
The stand basal area was not used as a predictor in Eq. 1 because growing stock was not measured in the plots at the time of the vegetation inventories in 2018 or 2022. The stand basal area removed in 2016 was tested as an additional continuous predictor in Eq. 1. In most cases, it was neither statistically significant nor did it change the effects of the year and treatment factors. In some cases, the correlation and confounding of the removed stand basal area with thinning intensity made it difficult to interpret the results. Thus, we used the treatment, measurement year, and their interaction as factors in two-way repeated measures ANOVA.
The models were fitted, and the marginal means were estimated using the UNIVARIATE procedure in IBM SPSS Statistics 29 (IBM SPSS Inc., Chicago, IL, USA). Post hoc least significant difference pairwise comparisons of estimated marginal means were performed for multiple comparisons.
3 Results
Based on statistical models, the effect of thinning intensity alone increased the coverage of a few species groups or species significantly: grasses and sedges, Betula pubescens Ehrh., Rubus idaeus, and Trientalis europaea L. (Table 2). Unlike those mentioned above, the abundance of T. europaea decreased a few years after harvest at both thinning intensities (Figs. 1, 2 and 4, Suppl. file S1).
| Table 2. Two-way repeated measures ANOVA with treatment (thinning intensity) and year (time since harvest) as fixed factors on the percent cover of species groups and species in the cutting trials on drained peatlands (Eq. 1). F-values and corresponding p-values indicated by asterisks (* p < 0.05, ** p < 0.01, *** p < 0.001) are presented only for the species groups and species with significant (p < 0.05) effects. For species abbreviations, see the text and Supplementary file S1. | ||||
| Species groups or species | Intercept | Treatment | Year | Treatment × Year |
| TREES and SHRUBS | 69.58** | 2.34 | 8.41*** | 3.20* |
| DWARF SHRUBS | 486.22*** | 0.29 | 37.33*** | 8.01*** |
| GRASSES and SEDGES | 10.66* | 4.12* | 34.13*** | 9.14*** |
| HERBS | 75.47** | 0.17 | 15.10*** | 5.51*** |
| BRYALES | 809.24*** | 1.72 | 17.20*** | 3.17* |
| SPHAGNUM | 302.12*** | 0.78 | 11.22*** | 1.21 |
| Betupube | 28.54* | 3.73* | 4.88* | 4.04** |
| Piceabie | 26.18* | 0.15 | 3.40* | 1.12 |
| Pinusylv | 7.43* | 2.32 | 5.85** | 1.84 |
| Rubuidae | 25.76* | 4.50* | 19.68*** | 5.80*** |
| Sorbaucu | 7.96 | 0.83 | 6.53** | 0.50 |
| Vaccmyrt | 82.98** | 0.15 | 49.48*** | 5.44** |
| Vaccviti | 36.19** | 1.14 | 9.01*** | 7.00*** |
| Careglob | 7.28 | 2.41 | 23.55*** | 6.40*** |
| Descflex | 5.37 | 0.84 | 6.08** | 1.02 |
| Dryocart | 185.00*** | 0.41 | 35.25*** | 2.73* |
| Epilangu | 15.31*** | 1.47 | 6.78** | 3.55* |
| Linnbore | 4.15 | 0.25 | 3.39* | 2.86* |
| Maiabifo | 3.45 | 0.33 | 7.61** | 0.46 |
| Melasylv | 1.85 | 2.37 | 3.05 | 2.85* |
| Oxalacet | 2.84 | 1.57 | 5.60** | 4.00** |
| Trieeuro | 10.76* | 4.50* | 17.10*** | 3.62* |
| Bracspp | 41.30* | 1.02 | 7.72** | 7.46*** |
| Dicrmaju | 26.18* | 2.94 | 26.32*** | 0.57 |
| Hylosple | 255.26*** | 0.39 | 14.82*** | 3.24* |
| Plagspp | 2.20 | 0.39 | 9.23*** | 1.46 |
| Pleuschr | 53.11** | 0.55 | 11.63*** | 3.63* |
| Sphagirg | 12.17* | 0.42 | 6.57** | 1.86 |
| Spharuss | 6.70 | 0.39 | 7.55** | 0.23 |
| Litter | 1088.50*** | 0.87 | 22.30*** | 2.85* |
| Number of species | 690.35*** | 0.42 | 4.17* | 2.95* |
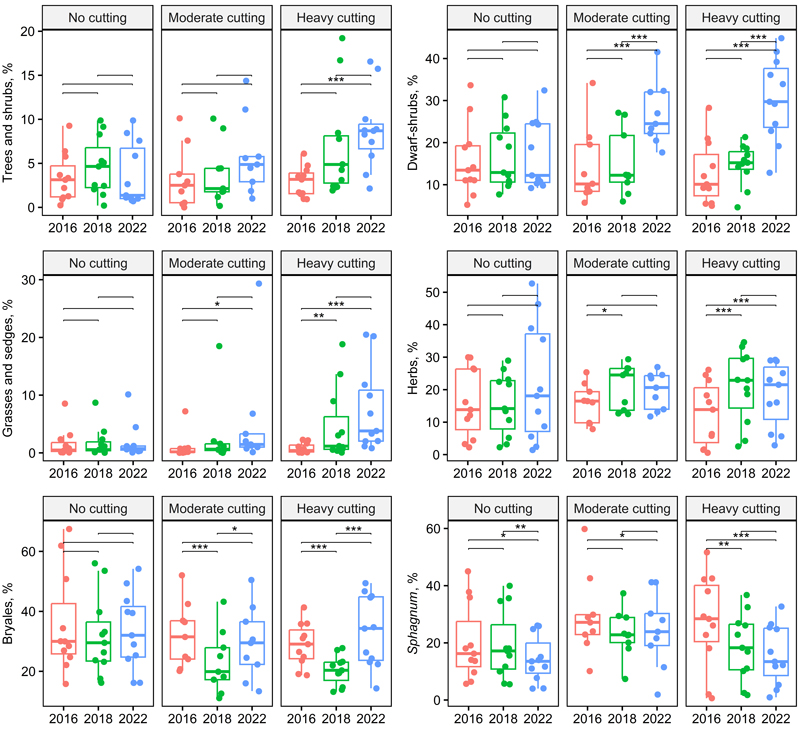
Fig. 1. Box plots showing species group cover across treatment and year (two-way repeated ANOVA, Table 2). Significant pairwise comparisons among years are indicated with asterisks (* p < 0.05, ** p < 0.01, *** p < 0.001). Treatments and stand basal area after cutting in 2016: no cutting (G = 20–30 m2 ha–1), moderate cutting (G = 17 m2 ha–1) and heavy cutting (G = 12–13 m2 ha–1).
For Pinus sylvestris L., Picea abies (L.) Karst., and Sorbus aucuparia L. seedlings, only the time since harvest was significant without an interaction (Table 2). The coverage of those species increased/decreased with the time after harvest but behaved similar to that between the harvesting intensities. The coverage of P. sylvestris was low in all years, while P. abies and S. aucuparia had the highest coverage 2 years after thinning on both control and thinned plots (Fig. 2, Suppl. file S1).
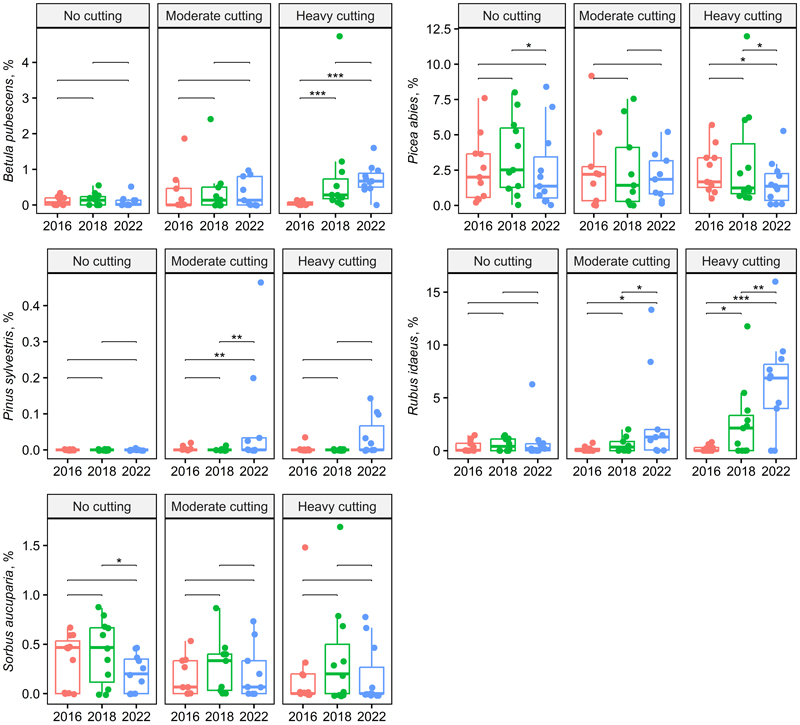
Fig. 2. Box plots showing the tree and shrub species cover across treatment and year (two-way repeated ANOVA, Table 2). Significant pairwise comparisons among years are indicated with asterisks (* p < 0.05, ** p < 0.01, *** p < 0.001). Treatments and stand basal area after cutting in 2016: no cutting (G = 20–30 m2 ha–1), moderate cutting (G = 17 m2 ha–1) and heavy cutting (G = 12–13 m2 ha–1).
However, there were several species and species groups that responded to the thinning intensity with an interaction showing a greater treatment effect with the more time since harvest. For example, the abundance of Carex globularis L., Dryopteris carthusiana (Vill.) H.P. Fuchs, Epilobium angustifolium L., Vaccinium myrtillus, V. vitis-idaea, Linnaea borealis L. and Brachythecium spp. increased more on intensively thinned plots, but that of Oxalis acetosella L. decreased (Table 2, Figs. 3 and 4). The abundance of V. myrtillus increased more than that of V. vitis-idaea, especially between 2018 and 2022 (Table 2, Fig. 3). Among dwarf shrubs, Linnaea borealis similarly responded to selection harvesting and time but to a lesser extent.
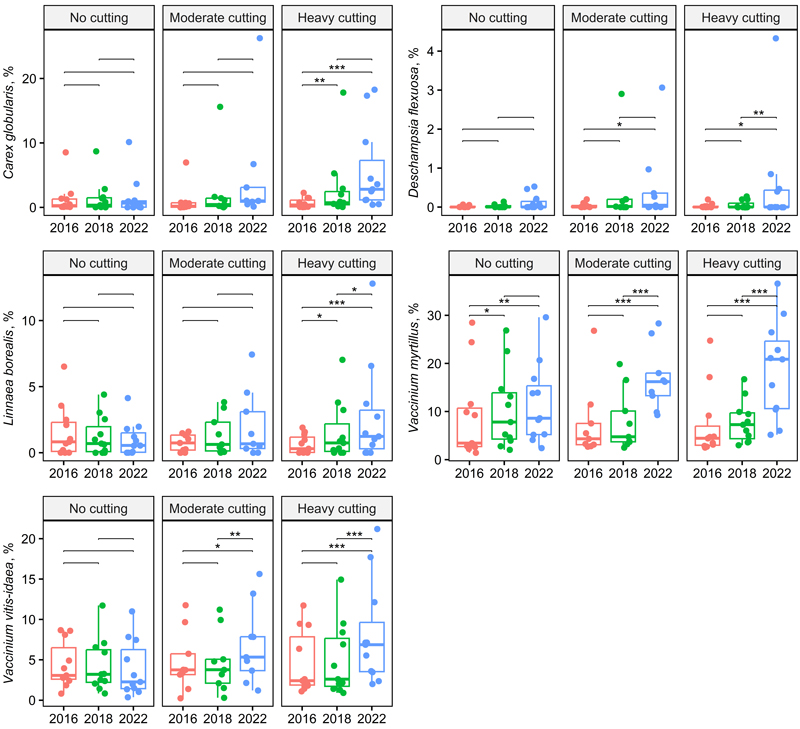
Fig. 3. Box plots showing grass and sedge and dwarf shrub species cover across treatment and year (two-way repeated ANOVA, Table 2). Significant pairwise comparisons among years are indicated with asterisks (* p < 0.05, ** p < 0.01, *** p < 0.001). Treatments and stand basal area after cutting in 2016: no cutting (G = 20–30 m2 ha–1), moderate cutting (G = 17 m2 ha–1) and heavy cutting (G = 12–13 m2 ha–1).
For Maianthemum bifolium (L.) F.W. Schmidt and Deschampsia flexuosa (L.) Trin, only the time effect was significant; their abundance slightly increased on both control and thinned plots, especially between 2018 and 2022 (Figs. 3 and 4). Melampyrum sylvaticum L. was very sparse and disappeared from thinned plots by 2022, which was 6 years after thinning; this occurred faster on intensively treated plots (Suppl. file S1).
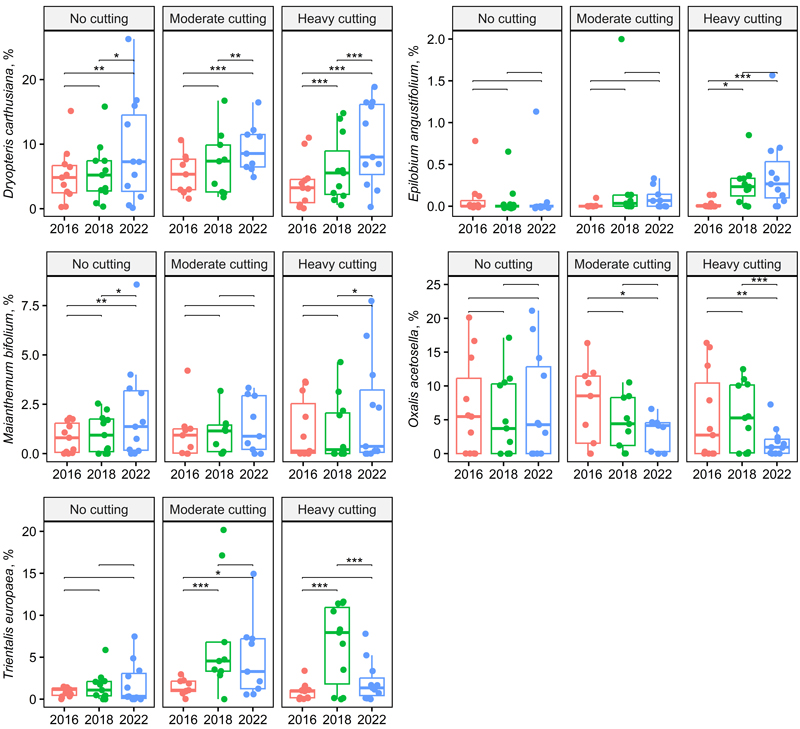
Fig. 4. Box plots showing herb species cover across treatment and year (two-way repeated ANOVA, Table 2). Significant pairwise comparisons among years are indicated with asterisks (* p < 0.05, ** p < 0.01, *** p < 0.001). Treatments and stand basal area after cutting in 2016: no cutting (G = 20–30 m2 ha–1), moderate cutting (G = 17 m2 ha–1) and heavy cutting (G = 12–13 m2 ha–1).
The abundance of Dicranum majus Sm. initially declined but then slightly increased on both control and treated plots; only the time effect was significant (Fig. 5, Table 2). Plagiothecium spp. were scarce, but its abundance slightly increased across all treatments (Suppl. file S1).
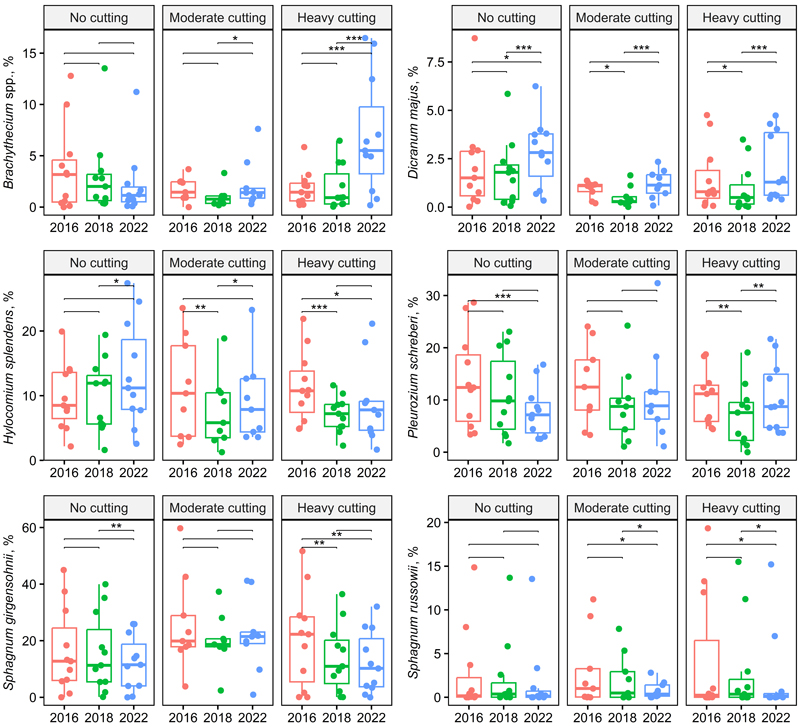
Fig. 5. Box plots showing the bryales and Sphagnum species cover across treatment and year (two-way repeated ANOVA, Table 2). Significant pairwise comparisons among years are indicated with asterisks (* p < 0.05, ** p < 0.01, *** p < 0.001). Treatments and stand basal area after cutting in 2016: no cutting (G = 20–30 m2 ha–1), moderate cutting (G = 17 m2 ha–1) and heavy cutting (G = 12–13 m2 ha–1).
The abundance of Hylocomium splendens (Hedw.) Schimp. slightly increased in control plots; in treated plots, it initially declined but began to increase by 2022, especially on less intensively thinned plots. The abundance of Pleurozium schreberi (Wild. ex Bird.) Mitt. declined in control plots and initially declined in treated plots but later began to recover (Fig. 5, Suppl. file S1).
As a group, the abundance of Sphagnum mosses decreased in both control and treated plots, with only the effect of time being significant (Table 2). Among species, S. girgensohnii Russow and S. russowii Warnst. followed the same pattern, showing declines (Fig. 5).
The litter coverage increased slightly in control plots but initially increased and showed a slight decline by 2022 in treated plots (Table 2, Suppl. file S1).
The thinning intensity did not have a significant effect on the number of species, but the number of species increased slightly on the thinned plots (Table 2, Suppl. file S1).
4 Discussion
4.1 Trees and shrubs
Thinning significantly increased the abundance of Betula pubescens seedlings and Rubus idaeus. They also reacted to the thinning intensity through an interaction showing a greater treatment effect with the more time since thinning. It is well known that the abundance of B. pubescens increases in the succession of drained peatland forests because of fertilisation and logging (Saarinen et al. 2020; Haapakoski et al. 2021; Väänänen et al. 2024). Rubus idaeus, which benefits from increased light exposure and soil disturbance (Ricard and Messier 1996; Vanha-Majamaa et al. 2017), rapidly became more abundant after thinning, particularly in the most intensively thinned plots. Rubus idaeus also benefits from increased soil nitrogen mobilisation following logging and the nutrients released from logging residues (Bråkenhielm and Liu 1998). Picea abies and Sorbus aucuparia seedlings were slightly more abundant 2 years after harvest. This is mainly because in 6 years (by 2022), the seedlings had grown taller than 0.5 m and, thus, no longer belonged to the field layer considered in this study. In contrast, the number of B. pubescens seedlings continued to increase as new seedlings emerged. The number of P. sylvestris seedlings in the spruce-dominated plots was expectedly very low (Saksa and Valkonen 2011).
4.2 Dwarf shrubs
As a group, dwarf shrubs and bilberry (Vaccinium myrtillus) exhibited the same pattern: increasing between 2018 and 2022 and increasing more on the most intensively thinned plots. Lingonberry (Vaccinium vitis-idaea) and Linnaea borealis followed this pattern less consistently. Previous studies have shown that bilberry and lingonberry benefit from thinning (Tonteri et al. 2016; Lõhmus and Remm 2017), but according to Turtiainen et al. (2013, 2016), this only occurs after a brief initial decline or delay. In the present study, the coverage of bilberry increased more than that of lingonberry after 2 years. The sites were also more favourable for bilberry than lingonberry (Salemaa 2001a, b). The abundance of L. borealis increases as forests become older, with its coverage peaking in 80–120-year-old forests, but it can also thrive in forest regeneration areas (especially in Northern Finland) on the shaded sides of ploughing furrows (Salemaa 2001c and references therein).
Bilberry coverage in peatlands is generally lower than that in mineral soils (Salemaa 2001a; Turtiainen et al. 2016). Among peatland site classes (Laine et al. 2018; Korhonen et al. 2024), bilberry coverage is highest in class III (bilberry-rich sites) and IV (lingonberry-rich sites) in spruce mires, site type IV (pine mires) and comparable peatland forests (Turtiainen et al. 2016). After the seedling stage, bilberry coverage typically increases up to a threshold as the stand ages and the basal area grows, after which the canopy density limits bilberry coverage. Considering different models, bilberry coverage reaches its peak when the stand basal area is between 20 and 30 m2 ha–1 (Miina et al. 2009; Turtiainen et al. 2016; Eldegard et al. 2019; Villén-Peréz et al. 2020; Haapakoski et al. 2021). The optimal basal area for bilberry is lower in spruce-dominated stands than in pine-dominated stands (Eldegard et al. 2019). When the forest is younger than 80 years old or dominated by Norway spruce or deciduous trees, the optimal stand density for bilberry is reduced to around 20 m2 ha–1. The results of the present study suggest even smaller basal areas in spruce stands on forest-drained peatlands.
In the moist conditions of the drained spruce peatlands in this study, bilberry showed no signs of decline following logging but rather an increase after a short delay. Bilberry’s linear decrease in cover with increasing felling intensity in mineral soil forests appears well established (Bergstedt and Milberg 2001; Vanha-Majamaa et al. 2017), as does its sharp decline and slow recovery after clear-felling combined with site preparation (Salemaa 2001a; Tonteri et al. 2016). According to Vanha-Majamaa et al. (2017), bilberry cover remains lower than the control even 10 years after logging and treatment methods, small-gap felling, and selection harvesting without soil preparation. This may be partly explained by the fact that bilberry abundance varies between years due to differing weather conditions. However, bilberry’s recovery is faster after less intensive treatments, e.g. after selection harvesting (Atlegrim and Sjöberg 1996; Rydgren et al. 2004; Vanha-Majamaa et al. 2017).
Hedwall et al. (2013) suggested lengthening the rotation period to favour bilberries. According to Vanha-Majamaa et al. (2017), less destructive site preparation methods and avoiding soil disturbance would also favour this species. The high morphological plasticity and rapid vegetative recovery of deciduous bilberry enable it to respond rapidly to changing environmental conditions (Tolvanen 1995; Tolvanen and Laine 1997), e.g. to typical and suitable light conditions for bilberry in selection harvesting sites of continuous cover forestry.
The optimal habitat for lingonberry is less fertile than that evaluated in this study (Salemaa 2001b; Turtiainen et al. 2013). Our results agree with previous studies that have shown that lingonberry withstands selection harvesting well; after more intensive cutting, its coverage usually decreases but recovers quickly (Vanha-Majamaa et al. 2017). Hamberg et al. (2019) observed the rapid recovery of lingonberry in drained areas. Lingonberry tolerates light and drought better than thin-leaved bilberry, which explains its faster recovery after regeneration felling in mineral soils (Salemaa 2001a, 2001b). However, bilberry responded more quickly and strongly than lingonberry to the thinning operations in moisture conditions of the drained sites in this study.
4.3 Grasses and sedges
As a group, grasses and sedges benefited from tree stand thinning. The coverage of this group increased more rapidly in the heavy cutting plots than in the moderate cutting plots. The abundance of Carex globularis increased more in areas with more intensive thinning. The coverage of C. globularis expands after thinning, at least temporarily, as light availability and soil moisture increase (Haapakoski et al. 2021), with its optimal growth sites being V. myrtillus and V. vitis-idaea drained peatlands (Hotanen 2001a). The occurrence of logging trails in some vegetation plots may explain the increase in C. globularis, as it benefits from disturbances, such as soil scarification, and thrives in patchy areas (Moilanen et al. 1995; Saarinen 2013).
Deschampsia flexuosa is common in drained spruce peatland forests and benefits from increased light availability (Mäkipää 2001a). Its abundance increased in the study plots, particularly in treated areas, but also slightly in control plots, with the only significant factor being the effect of time. This species also becomes more abundant with forest succession caused by drainage (Mäkipää 2001a), which occurred in the study plots, as evidenced by the decline in Sphagnum mosses.
4.4 Herbs
The herbaceous plant group includes species that either increase or decline rapidly after harvest due to changes in light availability (Hannerz and Hånell 1997; Tarvainen et al. 2015; Tonteri et al. 2016). After lighter harvesting intensities, changes in coverage are smaller than those following clear-cutting (Saarinen 2013; Vanha-Majamaa et al. 2017; Hamberg et al. 2019). Trientalis europaea and Dryopteris carthusiana immediately benefited from selection harvesting. These species tolerate light well and spread efficiently (Nousiainen 2001; Tonteri 2001a; Haapakoski et al. 2021); thus, their increase after harvest was expected, and similar changes can also be anticipated following conventional thinning.
The abundance of T. europaea declined between 2018 and 2022 in the treated plots. This species benefits from gaps in the field and bottom layers and is among the first species to colonise patches created by disturbances; most of its rapid spread is clonal (Hiirsalmi 1969; Piqueras and Klimes 1998). Its competitive advantage over other vegetation seems to disappear quickly (Hiirsalmi 1969). Trientalis europaea benefits from drainage (Tonteri 2001a) and showed slight increases in abundance in the control plots. According to Tonteri (2001a), T. europaea is most common and abundant in forests under 40 years old, although it can persist in older forests.
Among common fern species, Dryopteris carthusiana is prevalent in herb-rich and V. myrtillus peatland forests, but it is less common and scarce in V. vitis-idaea peatland forests. It benefits from forest drainage and fertilisation (Nousiainen 2001). Dryopteris carthusiana tolerates harvesting well and has previously been observed to increase rapidly after selection harvesting in spruce peatland forests (Haapakoski et al. 2021). Epilobium angustifolium, which is a light- and nitrogen-demanding species (Tamm 1956), responded as expected, showing a positive impact of time on abundance with the increase in the harvest intensity. In both mineral and peat soils, E. angustifolium is most common and abundant in tree-less and very young forests (Vanha-Majamaa 2001).
In the selection-harvested plots, especially in the more intensively thinned areas, Oxalis acetosella declined and grew in sheltered locations away from direct sunlight. This species thrives in herb-rich peatland forests in the southern half of Finland, herb-rich forests, and herb-rich heath forests (Tonteri 2001b; Laine et al. 2018). It is largely absent or very scarce in other peatland habitats. Oxalis acetosella is most abundant in sufficiently old (approximately 40–80 years), shady forests. Due to its thin leaves, it does not tolerate clear-cutting, but it can grow in openings and young forests, sometimes forming dense patches at the bases of large stones and stumps, where moist, sheltered microhabitats develop (Tonteri 2001b).
Maianthemum bifolium increased slightly in control and treated areas, with no interaction between thinning intensity and time. Moderately shaded forests are its optimal habitat, and its relatively thin leaves utilise light efficiently. The delicate root system of M. bifolium is close to the soil surface and is sensitive to drying, but this is less common in peatlands than in mineral soils. Shade is crucial for M. bifolium, as it can suffer frost damage in open patches (Tonteri 2001c and references therein). It is a weak competitor. In recently logged openings, it can spread and increase rapidly through asexual reproduction, but it declines quickly as the young stand develops (Tonteri 2001c).
The very scarce Melampyrum sylvaticum disappeared entirely from the harvested areas. The abundance of this annual species may be influenced by phenological fluctuations between years. It can grow in nutrient-rich mineral soil forests of various ages but is sparse in densely forested, heavily shaded areas. Melampyrum sylvaticum can occur, albeit rarely, on hummock surfaces in mesotrophic spruce mires, fens and meadows (Jalas 1980). There were relatively few observations on drained peatlands, but M. sylvaticum has been shown to increase slightly as peatland forest succession progresses after drainage (Sarasto 1961).
4.5 Bryales and Sphagnum
In the stable and shaded conditions of the control plots, the abundance of Hylocomium splendens increased slightly, while that of the more light-dependent Pleurozium schreberi declined somewhat (Mäkipää 2001b, 2001c). In the harvested areas, the abundance of both species initially declined but began to recover by 2022. Immediately after harvest, they were partially buried or smothered under increased litterfall. Hylocomium splendens does not tolerate direct sunlight, and its coverage can be low even in canopy openings (Mäkipää 2001b). Aa decline has been observed in mineral soils after small-gap and selection harvesting (Vanha-Majamaa et al. 2017). In peatlands, which are more moisture-retentive than mineral soils, H. splendens typically collapses following clear-cutting (Hamberg et al. 2019). Vanha-Majamaa et al. (2017) showed that the coverage of P. schreberi decreased after all treatments. However, it recovered quickly and was even more abundant after 10 years than before treatment, especially after selection harvesting. Pleurozium schreberi has a strong dispersal and growth capacity (Lloret 1994; Frego 1996; Salemaa et al. 2008), particularly when the soil surface is left undisturbed (Vanha-Majamaa et al. 2017).
The abundance of Brachythecium spp., which primarily live on litter and logging residues (Mäkipää 2001d), slightly increased in areas that were more intensively harvested. The litter coverage also increased more after intensive stand thinning compared to light thinning. Based on field observations, rainy summers also quickly increase the abundance of Brachythecium spp. Plagiothecium spp. include species that live on litter (Ulvinen et al. 2002), which may explain the slight increase in their abundance in control and thinned areas (litter coverage also increased in control areas).
Dicranum majus behaved similarly in both control and thinned areas, showing an initial decline but then an increase and narrowly surpassing its original abundance from 2016. The logging residues and increased litter in thinned areas may explain the temporary decline observed at treated sites. The temporary regression in control areas is harder to explain; although needle litter coverage also increased, it might be due to interannual weather variations. Dicranum majus is more nutrient demanding and grows in shadier habitats than Dicranum polysetum Sw. (Mäkipää 2001e, 2001f). Both species benefit from peatland drying and forest succession (Sarasto 1961). Dicranum polysetum is resilient to forest disturbances, including changes in light conditions and drought (Peterson and Mayo 1975; Tonteri et al. 2016), and showed no statistically significant changes in abundance. However, its abundance slightly increased in harvested areas but decreased in shadier control areas.
Some vegetation changes, such as the decline of Sphagnum (Hotanen 2001b), indicate that forest succession after drainage progressed in control and treated plots. Only the time since harvest was significant. Warm and dry summers during the study period 2016–2022 likely promoted the decline; also in the treated plots, where the water table is supposed to slightly rise. At the species level, this pattern was followed by the dominant species of the experimental plots, S. girgensohnii, and the slightly less demanding, common S. russowii.
Some pioneer species, such as Dicranella cerviculata (Hedw.) Schimp., which emerged in the tracks of heavy machinery but declined by 2022, were observed as new arrivals in the experimental plots. Dicranella cerviculata thrives on bare peat surfaces, and Saarinen et al. (2009) observed that it rapidly colonises peat patches when the water level is at a suitable depth.
The total number of species (field and ground layer together) increased slightly on the thinned plots over time. After harvest, some changes were attributed to immediate mechanical disturbances caused by logging, such as logging residues and machine tracks, while others result from alterations in light and moisture conditions (Brosofske et al. 1997). The effects of these factors were more pronounced in intensive treatments and affected both field- and ground-layer vegetation.
5 Conclusions
We verified our hypothesis that the harvest intensity, time since harvest, and their interaction affect vegetation dynamics on fertile drained Norway spruce peatlands. After harvesting, plant species respond to changed conditions. According to statistical models, some species and species groups reacted to thinning intensity alone, but several species and species groups responded to the thinning intensity through an interaction showing greater treatment effects with more time since thinning. For some species, only the time since harvest was significant. The effect of selection harvesting on the number of plant species seemed minor in drained Norway spruce peatlands. The effects of logging residues, strip roads and light availability may be the major drivers of the changes in species abundance. The effects of these factors are generally more pronounced with intensive treatments and apply to field- and ground-layer vegetation. Plant communities are resilient to selection harvesting, but the changes become clearer over time. The results could be generalised to the effects of conventional thinning; however, no studies have shown the effects of normal thinning on vegetation in drained peatland forests.
Authors’ contributions
The authors confirm contribution to the paper as follows:
J-PH; Methodology, conception and study design, Data collection: Analysis and interpretation of results, First draft, Writing—editing.
JM; Methodology, Analysis and interpretation of results, Writing—review and editing.
LK; Methodology, conception and study design, Data collection, review and editing.
RM; Conceptualisation, Funding acquisition, Project administration, Resources, Writing—review & editing.
Declaration of generative AI and AI-assisted technologies in the writing process
During the preparation of this work, the authors used DeepL to improve language. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the content of this publication.
Funding
This study received funding from European Union’s Horizon 2020 under Grant Agreement number 101000289 for the HoliSoils project and from the Flagship Program of Research Council of Finland for UNITE Flagship (decision no 359174).
Declaration of openness of research materials, data, and code
The data and SPSS syntax are available through the open research repository Zenodo: https://doi.org/10.5281/zenodo.18595336.
References
Äijälä O, Koistinen A, Sved J, Vanhatalo K, Väisänen P (eds) (2014) Metsänhoidon Suositukset. [Silvicultural Recommendations]. Metsätalouden kehittämiskeskus Tapion julkaisuja. Metsäkustanus Oy, Helsinki.
Atlegrim O, Sjöberg K (1996) Response of bilberry (Vaccinium myrtillus) to clear-cutting and single-tree selection harvests in uneven-aged boreal Picea abies forests. Forest Ecol Manag 86: 39–50. https://doi.org/10.1016/S0378-1127(96)03794-2.
Bergstedt J, Milberg P (2001) The impact of logging intensity on field-layer vegetation in Swedish boreal forest. Forest Ecol Manag 154: 105–115. https://doi.org/10.1016/S0378-1127(00)00642-3.
Bråkenhielm S, Liu Q (1998) Long-term effects of clear-felling on vegetation dynamics and species diversity in a boreal pine forest. Biodivers Conserv 7: 207–220. https://doi.org/10.1023/A:1008836502640.
Brosofske KD, Chen J, Naiman RJ, Franklin JR (1997) Harvesting effects on microclimate gradients from small stream to uplands in western Washington. Ecol Appl 7: 1188–1200. https://doi.org/10.2307/2641207.
Eldegard K, Scholten J, Stokland J, Granhus A, Lie M (2019) The influence of stand density on bilberry (Vaccinium myrtillus L.) cover depends on stand age, solar irradiation, and tree species composition. Forest Ecol Manag 432: 582–590. https://doi.org/10.1016/j.foreco.2018.09.054.
Frego KA (1996) Regeneration of four boreal bryophytes: colonization of experimental gaps by naturally occurring propagules. Can J Bot 74: 1937–1942. https://doi.org/10.1139/b96-231.
Haapakoski J, Hotanen J-P, Miina J, Korpela L, Mäkipää R (2021) Erirakenteishakkuiden vaikutus aluskasvillisuuden rakenteeseen metsäojitetuissa korvissa. [Short-term effects of selection harvesting on the structure of understorey vegetation in drained Picea abies mires]. Suo – Mires and peat 72(1): 1–27. https://www.suo.fi/volume/72.
Hamberg L, Hotanen J-P, Nousiainen H, Nieminen TM, Ukonmaanaho L (2019) Recovery of understorey vegetation after stem-only and whole-tree harvesting in drained peatland forests. Forest Ecol Manag 442: 124–134. https://doi.org/10.1016/j.foreco.2019.04.002.
Hämet-Ahti L, Suominen J, Ulvinen T, Uotila P, Vuokko S (1998) Retkeilykasvio. [Field Flora of Finland], 4th edition. Finnish Museum of Natural History, Botanical Museum, Helsinki. ISBN 951-45-8167-9.
Hannerz M, Hånell B (1997) Effects on the flora in Norway spruce forest following clearcutting and shelterwood cutting. For Ecol Manag 90: 29–49. https://doi.org/10.1016/S0378-1127(96)03858-3.
Hedwall P-O, Brunet J, Nordin A, Bergh J (2013) Changes in the abundance of keystone forest floor species in response to changes of forest structure. J Veg Sci 24: 296–306. https://doi.org/10.2307/23466918.
Heithecker TD, Halpern CB (2007) Edge-related gradients in microclimate in forest aggregates following structural retention harvest in western Washington. For Ecol Manag 248: 163–173. https://doi.org/10.1016/j.foreco.2007.05.003.
Hiirsalmi H (1969) Trientalis europaea L. A study of the reproductive biology, ecology and variation in Finland. Ann Bot Fenn 6: 119–173. https://www.jstor.org/stable/23724180.
Hökkä H, Mäkelä H (2014) Post-harvest height growth of Norway spruce seedlings in northern Finland peatland forest canopy gaps and comparison to partial and complete canopy removals and plantations. Silva Fenn 48, article id 1192. https://doi.org/10.14214/sf.1192.
Hökkä H, Repola J (2018) Pienaukkohakkuun uudistumistulos Pohjois-Suomen korpikuusikossa 10 vuoden kuluttua hakkuusta. [Regeneration results in small gaps on fertile peatlands in Northern Finland 10 years after cutting]. Metsätieteen aikakauskirja, article id 7808. https://doi.org/10.14214/ma.7808.
Holgén P, Hånell B (2000) Performance of planted and naturally regenerated seedlings in Picea abies-dominated shelterwood stands and clearcuts in Sweden. For Ecol Manag 127: 129–138. https://doi.org/10.1016/S0378-1127(99)00125-5.
Hotanen J-P (2001a) Carex globularis, Pallosara. In: Reinikainen A, Mäkipää R, Vanha-Majamaa I, Hotanen J-P (eds) Kasvit muuttuvassa metsäluonnossa. [Changes in the frequency and abundance of forest and mire plants in Finland since 1950]. Tammi, Jyväskylä, pp 164–165. ISBN 951-31-1963-7.
Hotanen J-P (2001b) Sphagnum, Rahkasammalet. In: Reinikainen A, Mäkipää R, Vanha-Majamaa I, Hotanen J-P (eds) Kasvit muuttuvassa metsäluonnossa. [Changes in the frequency and abundance of forest and mire plants in Finland since 1950]. Tammi, Jyväskylä, pp 266–267. ISBN 951-31-1963-7.
Jalas J (1980) Melampyrum sylvaticum L. – Metsämaitikka. In: Jalas J (ed) Suuri kasvikirja III. Suomen luonto. [Flora of Finland]. Otava, Helsinki, pp 539–540. ISBN 951-1-05293-4.
Korhonen KT, Räty M, Haakana H, Heikkinen J, Hotanen J-P, Kuronen M, Pitkänen J (2024) Forests of Finland 2019–2023 and their development 1921–2023. Silva Fenn 58, article id 24045. https://doi.org/10.14214/sf.24045.
Laine J, Vasander H, Hotanen J-P, Nousiainen H, Saarinen M, Penttilä T (2018) Suotyypit ja turvekankaat – kasvupaikkaopas. [Peatland types and transformed mires – guide for classification]. Metsäkustannus Oy, Helsinki. ISBN 978-952-338-036-3.
Leppä K, Hökkä H, Laiho R, Launiainen S, Lehtonen A, Mäkipää R, Peltoniemi M, Saarinen M, Sarkkola S, Nieminen M (2020a) Selection cuttings as a tool to control water table level in boreal drained peatland forests. Front Earth Sci 8, article id 576510. https://doi.org/10.3389/feart.2020.576510.
Leppä K, Korkiakoski M, Nieminen M, Laiho R, Hotanen J-P, Kieloaho A-J, Korpela L, Laurila T, Lohila A, Minkkinen K, Mäkipää R, Ojanen P, Pearson M, Penttilä T, Tuovinen J-P, Launiainen S (2020b) Vegetation controls of water and energy balance of a drained peatland forest: responses to alternative harvesting practices. Agr For Met 295, article id 108198. https://doi.org/10.1016/j.agrformet.2020.108198.
Lloret F (1994) Gap colonization by mosses on a forest floor: an experimental approach. Lindbergia 19: 122–128. https://www.jstor.org/stable/20149874.
Lõhmus A, Remm L (2017) Disentangling the effects of seminatural forestry on an ecosystem good: bilberry (Vaccinium myrtillus) in Estonia. For Ecol Manag 404: 75–83. https://doi.org/10.1016/j.foreco.2017.08.035.
Mäkipää R (2001a) Deschampisa flexuosa, Metsälauha. In: Reinikainen A, Mäkipää R, Vanha-Majamaa I, Hotanen J-P (eds) Kasvit muuttuvassa metsäluonnossa. [Changes in the frequency and abundance of forest and mire plants in Finland since 1950]. Tammi, Jyväskylä, pp 152–153. ISBN 951-31-1963-7.
Mäkipää R (2001b) Hylocomium splendens, Metsäkerrosammal. In: Reinikainen A, Mäkipää R, Vanha-Majamaa I, Hotanen J-P (eds) Kasvit muuttuvassa metsäluonnossa. [Changes in the frequency and abundance of forest and mire plants in Finland since 1950]. Tammi, Jyväskylä, pp 248–250. ISBN 951-31-1963-7.
Mäkipää R (2001c) Pleurozium schreberi, Seinäsammal. In: Reinikainen A, Mäkipää R, Vanha-Majamaa I, Hotanen J-P (eds) Kasvit muuttuvassa metsäluonnossa. [Changes in the frequency and abundance of forest and mire plants in Finland since 1950]. Tammi, Jyväskylä, pp 251–253. ISBN 951-31-1963-7.
Mäkipää R (2001d) Brachythecium-suku, Suikerosammalet. In: Reinikainen A, Mäkipää R, Vanha-Majamaa I, Hotanen J-P (eds) Kasvit muuttuvassa metsäluonnossa. [Changes in the frequency and abundance of forest and mire plants in Finland since 1950]. Tammi, Jyväskylä, pp 238–239. ISBN 951-31-1963-7.
Mäkipää R (2001e) Dicranum majus, Isokynsisammal. In: Reinikainen A, Mäkipää R, Vanha-Majamaa I, Hotanen J-P (eds) Kasvit muuttuvassa metsäluonnossa. [Changes in the frequency and abundance of forest and mire plants in Finland since 1950]. Tammi, Jyväskylä, pp 242–243. ISBN 951-31-1963-7.
Mäkipää R (2001f) Dicranum polysetum, Kangaskynsisammal. In: Reinikainen A, Mäkipää R, Vanha-Majamaa I, Hotanen J-P (eds) Kasvit muuttuvassa metsäluonnossa. [Changes in the frequency and abundance of forest and mire plants in Finland since 1950]. Tammi, Jyväskylä, pp 244–246. ISBN 951-31-1963-7.
McDonald JH (2014) Handbook of biological statistics, 3rd edition. Sparky House Publishing, Baltimore, Maryland.
Miina J, Hotanen J-P, Salo K (2009) Modelling the abundance and temporal variation in the production of bilberry (Vaccinium myrtillus L.) in Finnish mineral soil forests. Silva Fenn 43: 577–593. https://doi.org/10.14214/sf.181.
Moilanen M, Ferm A, Issakainen J (1995) Kuusen- ja koivuntaimien alkukehitys korven uudistamisaloilla. [Development of Norway spruce and birch seedlings in regeneration on spruce mires]. Metsätieteen aikakauskirja 2/1995: 115–130 https://doi.org/10.14214/ma.5950.
Moilanen M, Issakainen J, Vesala H (2011) Metsän uudistaminen mustikkaturvekankaalla – luontaisesti vai viljellen? [Forest regeneration in drained Vaccinium myrtillus peatland – natural or cultivated?]. Working Papers of the Finnish Forest Research Institute 192. https://urn.fi/URN:ISBN:978-951-40-2287-6.
Nieminen M, Hökkä H, Laiho R, Juutinen A, Ahtikoski A, Pearson M, Kojola S, Sarkkola S, Launiainen S, Valkonen S, Penttilä T, Lohila A, Saarinen M, Haahti K, Mäkipää R, Miettinen J, Ollikainen M (2018) Could continuous cover forestry be an economically and environmentally feasible management option on drained boreal peatlands? For Ecol Manag 424: 78–84. https://doi.org/10.1016/j.foreco.2018.04.046.
Nilsson M-C, Wardle DA (2005) Understory vegetation as a forest ecosystem driver: evidence from the northern Swedish boreal forest. Front Ecol Environ 3: 421–428. https://doi.org/10.1890/1540-9295(2005)003[0421:UVAAFE]2.0.CO;2.
Nousiainen H (2001) Dryopteris Carthusiana, Metsäalvejuuri. In: Reinikainen A, Mäkipää R, Vanha-Majamaa I, Hotanen J-P (eds) Kasvit muuttuvassa metsäluonnossa. [Changes in the frequency and abundance of forest and mire plants in Finland since 1950]. Tammi, Jyväskylä, pp 186–188. ISBN 951-31-1963-7.
Päivänen J, Hånell B (2012) Peatland ecology and forestry – a sound approach. University of Helsinki, Department of Forest Sciences Publications 3. ISBN 9789521045318.
Peterson WL, Mayo JM (1975) Moisture stress and its effect on photosynthesis in Dicranum polysetum. Can J Bot 53: 2897–2900. https://doi.org/10.1139/b75-318.
Piqueras J, Klimes L (1998) Demography and modelling of clonal fragments in the pseudoannual plant Trientalis europaea L. Plant Ecol 136: 213–227. https://doi.org/10.1023/A:1009753515163.
Ricard JP, Messier C (1996) Abundance, growth, and allometry of red raspberry (Rubus idaeus L.) along a natural light gradient in a northern hardwood forest. For Ecol Manag 81: 153–160. https://doi.org/10.1016/0378-1127(95)03643-1.
Rydgren K, Økland RH, Hestmark G (2004) Disturbance severity and community resilience in a boreal forest. Ecology 85: 1906–1915. https://doi.org/10.1890/03-0276.
Saarinen M (2013) Männyn kylvö ja luontainen taimettuminen vanhoilla ojitusalueilla – turvemaiden uudistamisen erityispiirteitä. [Artificial and natural seeding of Scots pine in old drainage areas – unique features of forest regeneration on peatlands.]. Dissertationes Forestales 164. https://doi.org/10.14214/df.164.
Saarinen M, Hotanen J-P, Alenius V (2009) Muokkausjälkien kasvillisuuden kehittyminen ojitettujen soiden metsänuudistamisaloilla. [Vegetation succession in prepared microsites in drained peatland forest regeneration areas]. Suo 60: 85–109. https://www.suo.fi/volume/60.
Saarinen M, Valkonen S, Sarkkola S, Nieminen M, Penttilä T, Laiho R (2020) Jatkuvapeitteisen metsänkasvatuksen mahdollisuudet ojitetuilla turvemailla. [Possibilities for continuous cover forestry in drained peatland forests]. Metsätieteen aikakauskirja, article id 10372. https://doi.org/10.14214/ma.10372.
Saksa T, Valkonen S (2011) Dynamics of seedling establishment and survival in uneven-aged boreal forests. For Ecol Manag 261: 1409–1414. https://doi.org/10.1016/j.foreco.2011.01.026.
Salemaa M (2001a) Vaccinium myrtillus, Mustikka. In: Reinikainen A, Mäkipää R, Vanha-Majamaa I, Hotanen J-P (eds) Kasvit muuttuvassa metsäluonnossa. [Changes in the frequency and abundance of forest and mire plants in Finland since 1950]. Tammi, Jyväskylä, pp 128–130. ISBN 951-31-1963-7.
Salemaa M (2001b) Vaccinium vitis-idaea, Puolukka. In: Reinikainen A, Mäkipää R, Vanha-Majamaa I, Hotanen J-P (eds) Kasvit muuttuvassa metsäluonnossa. [Changes in the frequency and abundance of forest and mire plants in Finland since 1950]. Tammi, Jyväskylä, pp 136–138. ISBN 951-31-1963-7.
Salemaa M (2001c) Linnaea borealis, Vanamo. In: Reinikainen A, Mäkipää R, Vanha-Majamaa I, Hotanen J-P (eds) Kasvit muuttuvassa metsäluonnossa. [Changes in the frequency and abundance of forest and mire plants in Finland since 1950]. Tammi, Jyväskylä, pp 120–121. ISBN 951-31-1963-7.
Salemaa M, Mäkipää R, Oksanen J (2008) Difference in the growth response of three bryophyte species to nitrogen. Environ Pollut 152: 82–91. https://doi.org/10.1016/j.envpol.2007.05.019.
Sarasto J (1961) On classification of peatlands drained for forestry purposes. Acta For Fenn 74. https://doi.org/10.14214/aff.7132.
Sarkkola S, Hökkä H, Ahti E, Nieminen M, Koivusalo H (2012) Depth of water table prior to ditch network maintenance is a key factor for tree growth response. Scand J For Res 27: 649–658. https://doi.org/10.1080/02827581.2012.689004.
Tamm CO (1956) The response of Chamaenerion angustifolium (L.) Scop. to different nitrogen sources in water culture. Physiol Plant 9: 331–337. https://doi.org/10.1111/j.1399-3054.1956.tb09010.x.
Tarvainen O, Hekkala A-M, Kubin E, Tamminen P, Tolvanen A (2015) Soil disturbance and early vegetation response to varying intensity of energy wood harvest. For Ecol Manag 348: 153–163. https://doi.org/10.1016/j.foreco.2015.04.001.
Tolvanen A (1995) Aboveground growth habits of two Vaccinium species in relation to habitat. Can J Bot 73: 465–473. https://doi.org/10.1139/b95-047.
Tolvanen A, Laine K (1997) Effects of reproduction and artificial herbivory on vegetative growth and resource levels in deciduous and evergreen dwarf shrubs. Can J Bot 75: 656–666. https://doi.org/10.1139/b97-073.
Tonteri T (2001a) Trientalis europaea, Metsätähti. In: Reinikainen A, Mäkipää R, Vanha-Majamaa I, Hotanen J-P (eds) Kasvit muuttuvassa metsäluonnossa. [Changes in the frequency and abundance of forest and mire plants in Finland since 1950]. Tammi, Jyväskylä, pp 226–227. ISBN 951-31-1963-7.
Tonteri T (2001b) Oxalis acetosella, Käenkaali. In: Reinikainen A, Mäkipää R, Vanha-Majamaa I, Hotanen J-P (eds) Kasvit muuttuvassa metsäluonnossa. [Changes in the frequency and abundance of forest and mire plants in Finland since 1950]. Tammi, Jyväskylä, pp 210–211. ISBN 951-31-1963-7.
Tonteri T (2001c) Maianthemum bifolium, Oravanmarja. In: Reinikainen A, Mäkipää R, Vanha-Majamaa I, Hotanen J-P (eds) Kasvit muuttuvassa metsäluonnossa. [Changes in the frequency and abundance of forest and mire plants in Finland since 1950]. Tammi, Jyväskylä, pp 200–201. ISBN 951-31-1963-7.
Tonteri T, Salemaa M, Rautio P, Hallikainen V, Korpela L, Merilä P (2016) Forest management regulates temporal change in the cover of boreal plant species. For Ecol Manag 381: 115–124. https://doi.org/10.1016/j.foreco2016.09.015.
Turtiainen M, Miina J, Salo K, Hotanen J-P (2013) Empirical prediction models for the coverage and yields of cowberry in Finland. Silva Fenn 47, article id 1005. https://doi.org/10.14214/sf.1005.
Turtiainen M, Miina J, Salo K, Hotanen J-P (2016) Modelling the coverage and annual variation in bilberry yield in Finland. Silva Fenn 50, article id 1573. https://doi.org/10.14214/sf.1573.
Ulvinen T, Syrjänen K, Anttila S (eds) (2002) Suomen sammalet – levinneisyys, ekologia, uhanalaisuus. [Bryophytes of Finland – distribution, ecology and red list status]. Finnish Environment Institute. Suomen ympäristö 560. http://hdl.handle.net/10138/40617.
Väänänen P, Rinne-Garmston K, Hultman J, Hotanen J-P, Hökkä H, Kattilakoski M, Laurén A, Lehtonen A, Mäkiranta P, Nieminen M, Ojanen P, Palvi A, Peltoniemi K, Sarkkola S, Stenberg L, Viitasaari T, Laiho R (2024) Hiilitase ja siihen vaikuttavat tekijät tuhkalannoitetuissa suometsissä (SuoHiTu). Loppuraportti. [Carbon balance and factors affecting it in ash-fertilized peatland forests (SuoHiTu-project). Final report]. Natural Resources Institute Finland (Luke), Helsinki.
Vanha-Majamaa I (2001) Epilobium angustifolium, Maitohorsma. In: Reinikainen A, Mäkipää R, Vanha-Majamaa I, Hotanen J-P (eds) Kasvit muuttuvassa metsäluonnossa. [Changes in the frequency and abundance of forest and mire plants in Finland since 1950]. Tammi, Jyväskylä, pp 189–190. ISBN 951-31-1963-7.
Vanha-Majamaa I, Shorohova E, Kushnevskaya H, Jalonen J (2017) Resilience of understorey vegetation after variable retention felling in boreal Norway spruce forest – a ten-year-perspective. For Ecol Manag 393: 12–28. https://doi.org/10.1016/j.foreco.2017.02.040.
Villén-Peréz S, Heikkinen J, Salemaa M, Mäkipää R (2020) Global warming will affect the maximum potential abundance of boreal plant species. Ecography 43: 801–811. https://doi.org/10.1111/ecog.04720.
Total of 69 references.

